|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Patai S. — The Chemistry of Organic Silicon Compounds (part 1-2) |
|
|
 |
| Предметный указатель |
Silenes structural parameters for 236—237
Silenes structure of 1045—1046
Silenes substituted silenes 119—124
Silenes synthesis of 1044—1045 1046—1070
Silenes thermal isomerization/fragmentation of 1070—1081
Silenes thermodynamic stability of 121—122
Silenes transient species 1046—1070
Silenes unimolecular reactions of 1073
Silenyl anion 203
Siletanes 695
Siletes 695
Silicate anions 282—289
Silicenium ions 184—193
Silicenium ions alkylsilicenium ions 188 485
Silicenium ions as electrophiles in cyclization 469—473
Silicenium ions hydroxy-substituted 188
Silicenium ions phosphino-substituted 187
Silicenium ions rearrangements involving 454—465
Silicenium ions silyl-substituted 190
Silicenium ions soivation of 190—191
Silicenium ions stabilities of 449
Silicenium ions substituted 187—190 448—450
Silicenium ions sulfur-substituted 188
Silicide clusters 1423
Silicocations see «Silyl cations”
Silicocene 162 642
Silicon analytical determination of 394—398
Silicon atomic properties of 70 230
Silicon carbide fibers 43 48 1224 1233—1234
Silicon clusters 102
Silicon clusters transition-metal derivatives 1423—1424
Silicon dihalides, PE spectra of 570—571
Silicon dioxide 139
Silicon electronegativity of 895
Silicon halides enthalpies of formation of 389
Silicon halides PE spectra of 602—607
Silicon hydride cations, stabilities of 446—448
Silicon hydrides as reducing agents 1267 1274—1276
Silicon hydrides enthalpies of formation of 389
Silicon hydrides GC retention times for 421 422
Silicon hydrides NMR spectra of 530 531
Silicon isotope used in NMR 521
Silicon manufacture of 19
Silicon monoxide, PE spectrum of 570
Silicon oxide 139
Silicon sources of 18
Silicon tetrahalides, PE spectra of 604—607
Silicon uses of element 19
Silicon-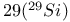 ESR/ENDOR spectroscopy 577 ESR/ENDOR spectroscopy 577
Silicon-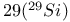 NMR spectroscopy 520—550 NMR spectroscopy 520—550
Silicon-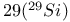 NMR spectroscopy chemical shift ranges listed 523 NMR spectroscopy chemical shift ranges listed 523
Silicon-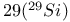 NMR spectroscopy electronegative substituent effect 524 NMR spectroscopy electronegative substituent effect 524
Silicon-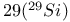 NMR spectroscopy organosilicon polymers 1309 NMR spectroscopy organosilicon polymers 1309
Silicon-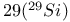 NMR spectroscopy paramagnetic relaxation reagent used 521 NMR spectroscopy paramagnetic relaxation reagent used 521
Silicon-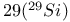 NMR spectroscopy polarization transfer used 521 522 NMR spectroscopy polarization transfer used 521 522
Silicon-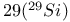 NMR spectroscopy polysilane polymers 1232—1233 NMR spectroscopy polysilane polymers 1232—1233
Silicon-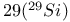 NMR spectroscopy reference compounds used 522—523 NMR spectroscopy reference compounds used 522—523
Silicon-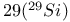 NMR spectroscopy solid-state NMR studies 544 NMR spectroscopy solid-state NMR studies 544
Silicon-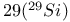 NMR spectroscopy spin-lattice relaxation 521 549—550 NMR spectroscopy spin-lattice relaxation 521 549—550
Silicon-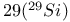 NMR spectroscopy spin-spin couplings 547—549 NMR spectroscopy spin-spin couplings 547—549
Silicon-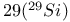 NMR spectroscopy transition-metal silyl complexes 1439 1440—1441 NMR spectroscopy transition-metal silyl complexes 1439 1440—1441
Silicon-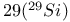 NMR spectroscopy “sagging” pattern of chemical shifts 524 NMR spectroscopy “sagging” pattern of chemical shifts 524
Silicon-bridged ring systems, photolysis of 981—984
Silicon-silicon bonds 249—254
Silicon-silicon bonds angles of 254
Silicon-silicon bonds dissociation energies of 385
Silicon-silicon bonds interactions, PE spectra affected by 587—593
Silicon-silicon bonds lengths of 249 251 1020
Silicon-silicon double bonds 234—236
Silicon-silicon double bonds length of 129—130
Silicon-X bonds bond dissociation energies of 77
Silicon-X bonds lengths of 71 74 1245
Silicon-X bonds strengths of 74—78
Silicon-X double bonds 103
Silicon-X multiple bonds in neutral compounds 481—483
Silicon-X multiple bonds ions containing 475—481
Siliconate anions, pentacoordinated form of 1265
Silicone oils, NMR spectra of 1318
Silicone polymers 31
Silicone polymers analysis of 410 413 415 419
Silicone polymers formation of 31 1013
Silicone polymers NMR spectra of 545—547 1318
Silicone polymers toxicological behavior of 1184
Silicones see also «Organofunctional siloxanes”
Siliconic acids 33
Siliconium ions, pentacoordinated 191
Silidyne groups, analysis of 406—407
Silines 698
Siliranes 694
Silirenes 694
Silkina, N.N. 483 (123) 508
Silolanes 697
Silole derivatives 696 697
Silolenes 696
Silolidines 718
Siloxane copolymers air-liquid interface behavior of 1354—1355
Siloxane copolymers composition of 1313—1314
Siloxane copolymers degradation of 1333—1335
Siloxane copolymers direct (thermodynamic) analysis of 1316—1319
Siloxane copolymers kinetic-chain analysis for 1315
Siloxane copolymers macrostructure of 1311—1314
Siloxane copolymers microstructure of 1314—1319
Siloxane copolymers number average sequence length of 1315—1316
Siloxane groupings, analysis of 415
Siloxane polymers degradation of 1319—1341
Siloxane polymers molecular weights of 1301
Siloxane polymers NMR spectra of 545—547 1318
Siloxane polymers redistribution reactions in 1301—1302
Siloxane polymers surface activity of 1351—1354 1355—1356
Siloxane polymers toxicological behavior of 1184
Siloxanes analysis of 394 395 398 406 408 409 418—419
Siloxanes basicity of 822
Siloxanes biological activity of 1154—1155
Siloxanes enthalpies of formation of 381—382
Siloxanes GC separation of 428—429
Siloxanes IR analysis of 418—419
Siloxanes NMR spectra of 534—536
Siloxanes ring-chain equilibria of 1292—1294
Siloxanes synthesis of 724—725 (see also “Cyclosiloxanes”; “Organofunctional siloxanes”; “Polysiloxanes”)
Siloxetenes, formation during photolysis 990
Siloxiranes, formation during photolysis 977
Siloxthianes 1402
Siloxy transfer 460
Siloxycarbenes, formation during photolysis 985
Silthianes cage silthianes 1401
Silthianes cyclic silthianes 1401—1403
Silthianes linear silthianes 1400—1401
Silver, J.M. 130 (209b) 220
Silver, S.M. 945 (267) 962
Silverman, G. 188 (316) 223 449 506
Silverman, M.A. 309 351 366
Silyamines, acidity of 814
Silyisilylenium ion 190
Silyl acetate 831
Silyl acetate derivatives 458
Silyl anion reagents 1420 1424
Silyl anions 201—204
Silyl anions barrier to inversion of 1008
Silyl anions configurational stability of 1008—1009
Silyl anions lone-pair conjugation in 1009—1010
Silyl anions NMR spectra of 514
Silyl anions nomenclature of 1007
Silyl anions PE spectrum of 1008
Silyl anions preparation of 450 1007—1008
Silyl anions pyramidal inversion of 1008
Silyl anions reaction with imines 951
Silyl anions reactivity of 495
Silyl anions structure of 1008
Silyl anions substituted 201—204
Silyl arenes, GC separation of 430
| Silyl arsines 1387—1389
Silyl bismuthines 1390
Silyl cations nomenclature of 1010
Silyl cations preparation of 1010—1011
Silyl cations structure of 1011—1013 (see also “Silylenium ions”)
Silyl chloride 180
Silyl chloride hydride attack on 208
Silyl enol ethers 732—737 1452 1467 1470
Silyl ester migration 469
Silyl esters as reagents 801—802
Silyl esters hydrolysis of 887—888
Silyl ethers basicity of 826
Silyl ethers GC separation of 429
Silyl ethers PE ionization energies of 636—639
Silyl fluoride 180
Silyl fluoride hydride attack on 208
Silyl Grignard reagents 330 1443
Silyl groups, electronic effects of 895—899
Silyl halides, PE ionization energies of 639—640
Silyl isocyanate 832 833
Silyl nitronates 830—831
Silyl peroxides, as reagents 802—803
Silyl radicals 198—200
Silyl radicals abstraction of hydrogen by 200
Silyl radicals PE spectra of 571
Silyl stibines 1389—1390
Silyl sulfides GC separation of 429
Silyl sulfides PE ionization energies of 636 638
Silyl thioacetate 832
Silyl thioethers, GC separation of 429
Silyl trioxides, as reagents 802
Silyl-nitrogen-X fragments, structural parameters of 258
Silyl-oxygen double bond, strength of 140
Silyl-oxygen-X fragments, structural parameters of 263
Silyl-substituted pentacoordinated silicon anions 206
Silyl-substituted phenyl cation 197
Silylacetylene 126 128 203
Silylacetylene radical cation, hyperconjugation in 593—596
Silylacetylenes see «Silynes”
Silylamine 79 180
Silylamines basicity of 817 819—822 825
Silylamines bond angles of 13
Silylamines enthalpies of formation of 382—383
Silylamines IR analysis of 421
Silylamines PE ionization energies of 628—631
Silylamines proton affinities of 820—822
Silylamines structural parameters of 254—256
Silylamines synthesis of 1515—1516
Silylated penta-3,4-diene-l-ynes 680
Silylation methods nitrogen-containing compounds 715
Silylation methods oxygen-containing compounds 742—744
Silylbarbarlane, photolysis of 981
Silylcarbenes formation during photolysis 989 990
Silylcarbenes siienes synthesized from 1060—1061
Silylcarbonylation 1518—1520
Silyldiazoalkanes, photolysis of 989—990
Silyldisilene 89 136 172
Silylene 167—169
Silylene addition to ethylene 182
Silylene complexes 42 180 183
Silylene complexes indirect evidence for 1427—1429
Silylene complexes synthesis attempted for 1429—1432
Silylene dissociation of 182
Silylene fluorosilane 180
Silylene heat of formation of 168—169
Silylene hydrogen 176—179
Silylene hydrogen abstraction by triplet silylene 181
Silylene hydrogen chloride 180
Silylene hydrogen fluoride 180
Silylene hydrogen sulfide 180
Silylene insertion into ammonia 180
Silylene ion 448
Silylene methane 178
Silylene phosphine 180
Silylene silanc 178
Silylene singlet-triplet energy difference 167—168
Silylene water 180
Silylenes 35 167—184
Silylenes cyclic 185
Silylenes dimerization of 175—176 977 1021 1027
Silylenes enthalpies of formation of 387
Silylenes expulsion from cyclic di-/tri-silenes 979
Silylenes formation during photolysis 978 998
Silylenes insertion reactions of 176—183
Silylenes ionic species 483
Silylenes multisilylenes 185
Silylenes noncyclic 185
Silylenes PE spectra of 563—568
Silylenes reactions of 175—183
Silylenes rearrangement to disilenes 1024—1025 1027—1029
Silylenes silenes synthesized from 1059—1060
Silylenes singlet-triplet energy differences 167—173
Silylenes stereochemistry of expulsion process 979—980
Silylenes substituent effects 169—170
Silylenes substituted silylenes 169—173
Silylenes synthesis of 976
Silylenes thermochemistry of 377
Silylenes UV-visible spectra of 173—175 (see also “Transition-metal silylene compounds”)
Silylenium cation 186
Silylenium cation heat of formation of 186—187
Silylenium ions 184—193
Silylenium ions heat of formation of 191
Silylenium ions preparation of 1010—1011
Silylenium ions structure of 1011—1013
Silylethylenes see «Silenes”
Silylethynes see «Silynes”
Silylidene 146—147
Silylisonitrile 80
Silyllithium derivatives 80 269 270 636 787—789 1425
Silylmercury derivatives 269 270—272 330 540 636 641 1440
Silylmethanol 80
Silylmethyl cation 194
Silylmethyl ketone 196
Silylmethylene 113
Silylnitrile 80
Silylolefins see «Silenes”
Silyloxazoles 690
Silyloxysilaethylene 119—123
Silyloxysilene 86
Silylphosphine 79 180
Silylphosphines acidity of 814
Silylphosphines reactions of 1365—1369
Silylphosphines structural parameters of 260
Silylphosphines synthesis of 1364
Silylphosphinometal derivatives 1384—1387
Silylpolyphosphines 1375—1376
Silylpolysulfides 1404
Silylpyridazines 693
Silylpyridines 692—693
Silylquinolizines 693—694
Silylsiiaethylene 119—123
Silylsilanone 140—141
Silylsilylene 131—133 172 178
Silynes 145—147 245
Silynes NMR spectra of 518
Silynes PE ionization energies of 620—622
Silynes synthesis of 677
Simchen, G. 682 693 734 754 759 800(152) 807
Simha, R.f. 1315 (141) 1359
Simhon, E.D. 1401 1408 1411
Simon, A. 48 (281) 56 246 249 296
Simon, G.L. 273 (346) 302
Simon, I. 419 (312) 440
Simonis, A.M. 1160 (143) 1202
Simons, J.H. 432 (411) 444
Simpson, C.C. 421 422 441
Simpson, J. 273 (344 345 349) 302 1418 1420 48 49) 1423 1434 134 135) 1435 1437 135) 1470 1471 1473
Simpson, K.A. 272 290 291 301 302 327 367 1437 1473
Simpson, P. 330 (97 99) 367
Simpson, R.N.F. 275 (369) 302 1434 1473
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



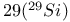 ESR/ENDOR spectroscopy
ESR/ENDOR spectroscopy