|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Carey F.A. — Organic Chemistry |
|
|
 |
| Предметный указатель |
Arylamines basicity of 920—922
Arylamines in reductive amination 934
Arylamines nomenclature of 913-916
Arylamines preparation of 932
Arylamines reactions of acylation 940—942
Arylamines reactions of electrophilic aromatic substitution 497 939—942 959
Arylamines reactions of nitrosation 945—950
Arylamines structure and bonding 916—918
Ascaridole 1104
Ascorbic acid (vitamin C) 54 827 1034 1055
Aspartame 1051—1052 1125
Aspirin 54
Aspirin inhibition of prostaglandin biosynthesis by 1083
Aspirin preparation of 1006—1008
Asymmetric center see “Chkality center”
Atactic polymers 313 610
Atomic number 7
Atomic number and the sequence rule 193
ATP see “Adenosine triphosphate”
Avery, Oswald 1166
Axial bonds in cyclohexane 117—120 135
Azeotropic mixture 638 722
Azide ion 31 327 328 338 347 349 779 927
Azo coupling 950—951 1004
Azo dyes 951—952
AZT see “Zidovudine”
Baeyer strain theory 112—113
Baeyer — Villiger oxidation 736—738 745 847
Barbiturates 900—901
Barton, Sir Derek 116
Base pairs 1166—1168
Base peak 569
Bases, used in elimination reactions 211—213 372—373 383 606
Basicity and nucleophilicity 348—350
Basicity constant 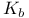 and and 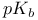 37—38 50 919 37—38 50 919
Basicity definition Arrhenius 33
Basicity definition Bronsted — Lowry 33
Basicity definition Lewis 45
Basicity of amines 919—923 955—956
Basicity of Grignard reagents 592—594 598
Basicity of heterocyclic amines 922—923
Basicity of leaving groups 330 352 944
Basicity of organolithium compounds 592—594
Becker, Luann 437
Beeswax 71 80 1079
Bender, Myron 852 855
Bending vibrations in infrared spectroscopy 559—560
Benedict’s reagent 1053—1054 1063
Benzal chloride 442
Benzaldehyde 432
Benzaldehyde diethyl acetal of 720
Benzaldehyde preparation of 711
Benzaldehyde reactions of Claisen — Schmidt condensation 775 783
Benzaldehyde reactions of nitration 498 927
Benzaldehyde reactions of reductive amination 935
Benzaldehyde reactions of with methylamine 724—726 927
Benzaldehyde reactions of with vinyllithium 597
Benzenamine 914 (see also “Aniline”)
benzene 58 424—431 463—464
Benzene,  of 37 of 37
Benzene, acidity of 593 621
Benzene, as industrial chemical 424
Benzene, Birch reduction of 438—439 440
Benzene, derivatives, nomenclature of 432—434
Benzene, electrophilic aromatic substitution in 475 table
Benzene, electrophilic aromatic substitution in bromination 475 480—481 504 505
Benzene, electrophilic aromatic substitution in chlorination 475 480
Benzene, electrophilic aromatic substitution in Friedel — Crafts acylation 475 484—488 504 505
Benzene, electrophilic aromatic substitution in Friedel — Crafts alkylation 475 481—483 510
Benzene, electrophilic aromatic substitution in nitration 475 477—478 505
Benzene, electrophilic aromatic substitution in sulfonation and disulfonation 475 478—480 500
Benzene, electrostatic potential map 423
Benzene, heat of hydrogenation 428—429
Benzene, isolation and discovery 424
Benzene, mass spectrum 568—569
Benzene, molecular orbitals 430—431 452—453
Benzene, nuclear shielding in 529
Benzene, stability of 428—429 463
Benzene, structure and bonding 424—428
Benzene, structure and bonding Kekule formulation 424—427 463
Benzene, structure and bonding orbital hybridization model 430
Benzene, structure and bonding resonance description 427—428
Benzenecarbaldehyde see “Benzaldehyde”
Benzenecarboxylic acid see “Benzoic acid”
Benzenediazonium chloride 946 1004
Benzenediols 994 (see also “Hydroquinone”; “Pyrocatechol”; “Resorcinol”)
Benzenesulfonic acid preparation of 475 478—480
Benzenesulfonic acid reactions of 500 1000
Benzimidazole 461
Benzofuran 460
Benzoic acid 424 432 792
Benzoic acid acidity of 803
Benzoic acid by oxidation of toluene 444
Benzoic acid esterification of 638 810—813
Benzonitrile 832
Benzophenone 706
Benzothiophene 460
Benzotrichloride 442
Benzoyl chloride 500 839 840
Benzoyl peroxide 442
Benzo[a]pyrene 435
Benzyl alcohol 711
Benzyl alcohol 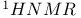 spectrum 545 spectrum 545
Benzyl bromide 434
Benzyl cation 438 445 571
Benzyl chloride, nucleophilic substitution in 672 784 808 841
Benzyl chloride, preparation of 442
Benzyl chloride, reaction of with lithium dimethylcuprate 617
Benzyl chloride, reaction of with magnesium 615
Benzyl chloride, reaction of with N-potassiophthalimide 930
Benzyl group 434
Benzyl radical 438 439 441—442
Benzylamine, preparation of 930
Benzylic 439
Benzylic halides, nucleophilic substitution in 444—446
Benzylic halogenation 439 441—443 466
Benzylic hydrogens 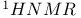 chemical shifts of 529 chemical shifts of 529
Benzyloxycarbonyl protecting group in peptide synthesis 1137—1139 1151
Benzyne as intermediate in nucleophilic aromatic substitution 981—985
Benzyne bonding in 982 984
Benzyne Dieis — Alder reactions of 985—986
Benzyne electrostatic potential map 984
Benzyne generation of 983 985—986 987
Berg, Paul 1181
Bergstrom, Sune 1084
Berthelot, Pierre — Eugene Marcellin 363
Berzelius, Jons Jacob 1—2 23
Bicarbonate 45 805
Bicarbonate  of 36 of 36
Bicyclic ring systems 130—131 136
Bicyclic ring systems as products in Dieis — Alder reactions 411 986
Big-bang theory 6
Bile acids and bile salts 1097—1098 1103
Bimolecular elementary step 154 158 164
Bimolecular elimination 214—217 (see also “E2 mechanism”)
Bimolecular nucleophilic substitution see “ mechanism” mechanism”
Bioenergetics 1162—1164
Biological isoprene unit see “Isopentenyl pyrophosphate”
Biosynthesis of amino acids, by transamination 1123—1125
Biosynthesis of cholesterol 1093—1095
Biosynthesis of ethylene 189
Biosynthesis of fatty acids 1075—1077
Biosynthesis of organohalogen compounds 767
Biosynthesis of phenols 1001—1002
Biosynthesis of prostaglandins 1081
Biosynthesis of terpenes 1087—1093
Biot, Jean Baptiste 287
Biphenyl 434 497 517
Birch reduction 438—439 440 464—465
Birch, Arthur J. 439
| Bisabolene 1104
Bloch, Felix 522
Bloch, Konrad 1093—1094
Boat conformation of cyclohexane 116—117 134
BOC see “tert-Butoxycarbony”
Boiling points and intermolecular attractive forces 80—83 147—150 708
Boiling points and intramolecular hydrogen bonds 996
Boiling points of alcohols 147—149 179 846
Boiling points of alkanes 63 80—83 846
Boiling points of alkyl halides 147—150 179
Boiling points of amines 918—919
Boiling points of carboxylic acids 794
Boiling points of esters 846
Boiling points of thiols 649
Bond angles acetaldehyde 706
Bond angles acetone 706
Bond angles acetylene 365—366 367
Bond angles ammonia 31
Bond angles and electron-pair repulsions 29
Bond angles aniline 916
Bond angles benzene 427
Bond angles boron trifluoride 31
Bond angles carbon dioxide 31
Bond angles cyclohexane 116
Bond angles cyclopropane 113 114
Bond angles dialkyl ethers 667
Bond angles enol of 2,4-pentanedione 762
Bond angles ethane 64 367
Bond angles ethylene 38—40 90 191 367
Bond angles ethylene oxide 667
Bond angles formaldehyde 31 706
Bond angles formic acid 793
Bond angles methane 31 64—65
Bond angles methanol 146—147 667 994
Bond angles methylamine 916 917
Bond angles phenol 994
Bond angles water 31 667
Bond angles [10]annulene 454
Bond dissociation energy 12 169—172 174
Bond dissociation energy 2-methylpropane 170—171 439
Bond dissociation energy acetylene 367
Bond dissociation energy and halogenation of methane 174
Bond dissociation energy aryl halides 972
Bond dissociation energy benzene 972
Bond dissociation energy ethane 169 170 367 972
Bond dissociation energy ethyl halides 972
Bond dissociation energy ethylene 191 367 972
Bond dissociation energy peroxides 242
Bond dissociation energy propane 169 170
Bond dissociation energy propene 395 439
Bond dissociation energy table 170
Bond dissociation energy vinyl halides 972
Bond distances 1,3-butadiene 400
Bond distances acetic acid 797
Bond distances acetylene 92 365—366 367
Bond distances alkyl halides 146
Bond distances allene 402
Bond distances ammonium acetate 797
Bond distances and strain 111 132
Bond distances benzene 427
Bond distances carbon-chlorine 834
Bond distances cyclobutadiene 451
Bond distances cyclooctatetraene 450
Bond distances dimethyl ether 667
Bond distances enol of 2,4-pentanedione 762
Bond distances ethane 64 367
Bond distances ethyl chloride 972
Bond distances ethylene 90 191 367
Bond distances ethylene oxide 667
Bond distances formic acid 793
Bond distances methane 63
Bond distances methanol 146
Bond distances methylamine 916 917
Bond distances phenol 994
Bond distances propene 191 367
Bond distances propyne 367
Bond distances vinyl halides 972
Bond lengths see “Bond distances”
Bond-line formulas 21 68 191
Bonding in  -unsaturated aldehydes and ketones 775-777 -unsaturated aldehydes and ketones 775-777
Bonding in acetylene 14 92—93 99 365—367 382
Bonding in alcohols 146—147
Bonding in aldehydes and ketones 706—708 742
Bonding in alkenes 89—92 99 190—192 220
Bonding in alkyl halides 146—147
Bonding in alkynes 365—367 382
Bonding in allene 402—404
Bonding in amines 916—918
Bonding in aryl halides 971—972
Bonding in benzene 427—428 430—431 452—453
Bonding in benzyne 982 984
Bonding in carbocations 160—162
Bonding in carboxylic acid derivatives 833—836
Bonding in carboxylic acids 793—794
Bonding in conjugated dienes 400
Bonding in ethane 67 95
Bonding in ethers and epoxides 667
Bonding in ethylene 14 89—91 99 190—191
Bonding in formaldehyde 14 706
Bonding in free radicals 167—168
Bonding in hydrogen 12 58—63
Bonding in methane 13 63—65
Bonding in phenols 994—995
Bonding models, comparison of 63 92—93 99
Bonds  in acetylene 92—93 99 365—366 in acetylene 92—93 99 365—366
Bonds  in ethylene 89—92 99 190—191 220 in ethylene 89—92 99 190—191 220
Bonds  in formaldehyde 706 in formaldehyde 706
Bonds axial and equatorial 117—120 135
Bonds bent, in cyclopropane 114
Bonds carbon-metal 587—589
Bonds covalent 12—13
Bonds double 14 191 220
Bonds hydrogen bonds 148 150—151 668
Bonds ionic 10—12
Bonds partial 155
Bonds polar  in acetylene 92—93 99 365—366 in acetylene 92—93 99 365—366
Bonds polar  in ethylene 89—92 99 190—191 220 in ethylene 89—92 99 190—191 220
Bonds polar  in methane and alkanes 63—65 67 in methane and alkanes 63—65 67
Bonds polar covalent 14—17
Bonds polar dipole moments of 17 table
Bonds polar three-center two-electron 252
Bonds polar triple 14 365—366
Borane 251
Borneol 1090
Borodin, Aleksandr 769
Borohydride ion 19 (see also “Sodium borohydride”)
Boron trifluoride Lewis acid/base complex with diethyl ether 46
Boron trifluoride VSEPR and molecular geometry of 30
Bradykinin 1135
Branched-chain carbohydrates 1043
Brevicomin 748
Broadband decoupling 553
Bromination 3-benzyl-2,6-dimethylphenol 1004
Bromination 4-chlor o-N-me thylaniline 503
Bromination aniline 497 950
Bromination anisole 495
Bromination benzene 475 480—481 504
Bromination benzylic, of alkylbenzenes 442—443 466
Bromination electrophilic aromatic substitution acetophenone 504
Bromination m-fluorophenol 1002
Bromination nitrobenzene 500 973
Bromination of aldehydes 757—759
Bromination of alkanes 177—178 180
Bromination of alkenes electrophilic 254—259 273 307—310 447-448
Bromination of alkenes free-radical 396—398 416
Bromination of alkynes 381
Bromination of benzene 475 480—481
Bromination of carboxylic acids 815—816 823
Bromination of conjugated dienes 407
Bromination of ketones 757—759 782
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



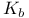 and
and 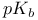
 of
of 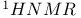 spectrum
spectrum  mechanism”
mechanism” -unsaturated aldehydes and ketones
-unsaturated aldehydes and ketones in acetylene
in acetylene in acetylene
in acetylene