|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Carey F.A. — Organic Chemistry |
|
|
 |
| Предметный указатель |
Structural formulas Fischer projections 293—295 316—317 1027—1028 1031 1061 1115—1116 1150
Structural formulas Lewis dot structures 12
Structural formulas Newman projections 105—106 109
Structural formulas of organic molecules 21—23
Structural formulas sawhorse 105—106
Structural formulas wedge-and-dash 29 105—106
Structural isomers see “Constitutional isomers”
Structural theory 3
Styrene 432
Styrene addition of bromine 447
Styrene addition of hydrogen bromide 448 466
Styrene industrial preparation of 269 424 446 483
Styrene polymers 270 449 1141—1142
Styrene polymers copolymer with 1,3-butadiene 408
Substituent effects (see also “Field effect”; “Inductive effect”; “Steric effects”)
Substituent effects on acidity of carboxylic acids 801—804
Substituent effects on acidity of phenols 998—999
Substituent effects on basicity of amines 919—923
Substituent effects on equilibrium, hydration of aldehydes and ketones 712—717
Substituent effects on rate and regioselectivity in electrophilic aromatic substitution 488—508 980
Substituent effects on rate of acid-catalyzed hydration 249
Substituent effects on rate of bimolecular nucleophilic substitution 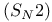 334—336 356 334—336 356
Substituent effects on rate of bromine addition to alkenes 259
Substituent effects on rate of epoxidation 262
Substituent effects on rate of nucleophilic aromatic substitution 975—980
Substituent effects on rate of unimolecular elimination 217—218
Substituent effects on rate of unimolecular nucleophilic substitution 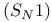 162—163 339—342 356 391—392 445 162—163 339—342 356 391—392 445
Substituent effects on stability of aldehydes and ketones 708
Substituent effects on stability of alkenes 197—200 221
Substituent effects on stability of carbocations 160—163 181 392 444—446
Substituent effects on stability of carbon-carbon triple bonds 374
Substituent effects on stability of free radicals 167—172 181 439 441
Substitution reactions 46 153—165 326—362
Substitution reactions allylic free radical 396—398 416
Substitution reactions allylic nucleophilic 393—394 416
Substitution reactions benzylic free radical 439 441—443 466
Substitution reactions benzylic nucleophilic 444—446 465
Substitution reactions electrophilic aromatic 473—518
Substitution reactions nucleophilic acyl 830—885
Substitution reactions nucleophilic aliphatic 160—161 163—165 326—362
Substitution reactions nucleophilic aromatic 975—985 986—987 1009
Substitution reactions of aryl diazonium salts 946—950 960—961
Substitutive nomenclature 144—145 178
Succinic acid 202 862
Succinic anhydride 486 862
Succinimide 397 443 862
Sucralose 1051—1052
Sucrose 1027 1048 1054
Sucrose octaacetate 1064
Sulfa drugs 951—952
Sulfanilamide 951
Sulfenic acids 650
Sulfhydryl group 648
Sulfides alkylation of 686—688 695
Sulfides oxidation of 685—686 695
Sulfides preparation of 650 685 694—695
Sulfinic acids 650
Sulfonate esters nucleophilic substitution reactions of 350—353 357
Sulfonate esters preparation of 351 357 636
Sulfonation of 1,2,4,5-tetramethylbenzene 510
Sulfonation of 2,6-dimethylphenol 1003
Sulfonation of benzene 475 478-480
Sulfonation of benzenesulfonic acid 500
Sulfones 686 695
Sulfonic acids 351 475 650
Sulfonium salts 686—688 695
Sulfoxides (see also “Dimethyl sulfoxide as solvent”)
Sulfoxides optically active 314
Sulfoxides preparation of 685—686 695
Sulfur trioxide 479
Sulfuric acid (see also “Sulfonation”)
Sulfuric acid  of 36 of 36
Sulfuric acid as catalyst for alcohol dehydration 203
Sulfuric acid as catalyst for dimerization of alkenes 266—267
Sulfuric acid as catalyst for Fischer esterification 638
Sulfuric acid as catalyst for hydration of alkenes 247—250 272
Sulfuric acid as catalyst for nitration of arenes 478
Sulfuric acid esters of 641
Sulfuric acid, addition to alkenes 245—247 272
Supercoiled DNA 1170—1172
syn Periplanar 217
Syndiotactic polymer 313—314 318
Synthon 895
Systeme International d’Unitds see “SI units”
Talaromycin A 748
Tariric acid 364
Tartaric acids 310
Tautomerism see “Keto-enol tautomerism”
Teflon 14 270
Terephthalic acid see “1 acid”
Termination step 173
Terpenes 1084—1093 1102
Terpenes and isoprene rule 1084—1086
Terpenes biosynthesis of 1087—1093
Terpenes classification 1085
tert-Butoxycarbonyl, protecting group in peptide synthesis 1138—1139 1143 1151
tert-Butyl alcohol see also “2-Methyl-2-propanol”
tert-Butyl alcohol, acidity of 37
tert-Butyl alcohol, dehydration of 203 207
tert-Butyl alcohol, esterification of 656 839
tert-Butyl alcohol, reaction with hydrogen chloride 152 153—160
tert-Butyl bromide, nucleophilic substitution in 339—341
tert-Butyl cation, electrostatic potential map 142
tert-Butyl cation, intermediate in acid-catalyzed hydration of 2-methylpropene 248
tert-Butyl cation, intermediate in dehydration of tert-butyl alcohol 207
tert-Butyl cation, intermediate in Friedel — Crafts alkylation of benzene 481—483
tert-Butyl cation, intermediate in nucleophilic substitution 339—341
tert-Butyl cation, intermediate in reaction of tert-butyl alcohol with hydrogen chloride 154 156—159
tert-Butyl cation, stability of 160
tert-Butyl chloride see also “2-Chloro-2-methylpropane”
tert-Butyl chloride by chlorination of 2-methylpropane 177
tert-Butyl chloride in Friedel — Crafts reaction 475 481—483
tert-Butyl chloride preparation from tert-butyl alcohol 152 153—160
tert-Butyl chloride reaction with lithium 589
tert-Butyl chloride solvolysis of 345—346 391
tert-Butyl group 74 (see also “1 group”)
tert-Butyl group large size of 123 124 128—129 200 334—335
tert-Butyl hydroperoxide 634—635 654
tert-Butyl methyl ether 672
tert-Butyl radical 168 171
tert-Butyl, alcohol,  of 37 40 of 37 40
tert-Butylcyclohexane, conformations 124
tert-Butyllithium 589
tert-Butyloxonium ion intermediate in hydration of 2-methylpropene 248
tert-Butyloxonium ion intermediate in hydration of dehydration of tert-butyl alcohol 207
tert-Butyloxonium ion intermediate in hydration of hydrolysis of tert-butyl bromide 339—341
tert-Butyloxonium ion intermediate in hydration of reaction of tert-butyl alcohol with hydrogen chloride 154—157 159—160
Tertiary carbon 74
Tertiary structure 1145—1148
Tesla unit of magnetic field strength 522
Tesla, Nikola 522
Testosterone 1100
Tetrachloromethane 150 166—167
Tetrafluoroethylene 14
Tetrafluoromethane 13
Tetrahedral geometry and 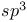 hybridization 64—65 hybridization 64—65
Tetrahedral geometry and VSEPR 29—31 49
Tetrahedral intermediate 811 831 837
Tetrahedral intermediate Claisen condensation 888
Tetrahedral intermediate Dieckmann condensation 890
Tetrahedral intermediate Fischer esterification 812—813 823
Tetrahedral intermediate in hydrolysis of acyl chlorides 840—841
Tetrahedral intermediate in hydrolysis of amides 864 866
Tetrahedral intermediate in hydrolysis of carboxylic acid anhydrides 844—845
Tetrahedral intermediate in hydrolysis of esters 850—852 856 876
Tetrahedral intermediate in reaction of esters with ammonia 858
Tetrahydrofuran 132 666
Tetrahydrofuran as solvent 591
Tetrahydrofuran, acid-catalyzed cleavage 676
Tetrahydrofuran, complex with borane 251
| Tetrahydrofuran, dipole moment of 668
Tetrahydropyran 666 667
Tetrahymanol 1104
Tetramethylsilane 525 526 549
Tetramethylsilane, electrostatic potential map 519
Tetrapeptide 1109
Tetraterpene 1085
Thalidomide 296
Theobromine 1158
Thermochemistry 86
Thermodynamic control addition of hydrogen bromide to 1,3-butadiene 407
Thermodynamic control addition to  unsaturated aldehydes and ketones 777—778 unsaturated aldehydes and ketones 777—778
Thermodynamic control Fries rearrangement 1006
Thermodynamic control glycoside formation 1046
Thermodynamic control Kolbe — Schmitt reaction 1006—1008
Thiazole 461
Thiirane 666
Thioesters acetyl coenzyme A 1070—1071
Thioesters nucleophilic acyl substitution in 835 858—859
Thiols acidity of 36 649 655 685
Thiols conjugate addition to  unsaturated carbonyl compounds 778 unsaturated carbonyl compounds 778
Thiols NMR spectra of 652
Thiols oxidation of 650—651 657
Thiols physical properties of 648—649
Thiols preparation of 648
Thionyl chloride 19
Thionyl chloride, reactions of carboxylic acids 485 810 838
Thionyl chloride, reactions of with alcohols 165 180 636
Thiopental sodium 901
Thiophene 460
Thiophene bonding in 463
Thiophene, electrophilic aromatic substitution in 508
Thiourea 901
Threo, stereochemical prefix 301—302
Thromboxanes 1081—1082
Thymidine 1159
Thymine 1157 1166
Thymol 1001
Thyroxine 275 297 974
Tin, reduction of nitro groups by 932 957
Toluene 423 424
Toluene benzylic halogenation of 442
Toluene bond dissociation energy 439
Toluene nitration of 488—492 506
Toluene oxidation of 444
Toluene physical properties of 995
Torsion angle 105—107
Torsional strain 107 133
Torsional strain, boat conformation of cyclohexane 116—117
Torsional strain, cyclobutane 115
Torsional strain, cyclopentane 115
Torsional strain, cyclopropane 114
Torsional strain, eclipsed conformation of butane 109—110 112
Torsional strain, eclipsed conformation of ethane 107
Tosylates see “p-Toluenesulfonic acid esters”
trans-Cycloheptene 201
Transamination 1123—1125
Transcription 1172 1174—1175 1188
Transfer RNA see “Ribonucleic acid transfer”
Transition metal organometallic compounds 608—610 616—617
Transition state addition of bromine to alkenes 259
Transition state and activation energy 108
Transition state bimolecular elimination (E2) 214—215
Transition state bimolecular nucleophilic substitution 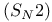 163—165 331 333 354 356 163—165 331 333 354 356
Transition state bimolecular nucleophilic substitution 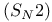 electrostatic potential map 326 electrostatic potential map 326
Transition state bond rotation in ethane 108
Transition state carbocation rearrangement 209—210
Transition state conversion of primary alcohols to primary alkyl halides 163—165 181 354
Transition state Dieis — Alder reaction 409
Transition state double-bond rotation 193
Transition state epoxide ring opening 680 681—682
Transition state free-radical halogenation 176
Transition state nucleophilic capture of carbocation 158 159 341
Transition state oxonium ion dissociation 156—157 159 162—163
Transition state proton transfer 155 159
Transition state unimolecular nucleophilic substitution  156—159 341 156—159 341
Translation 1172 1177 1178—1179
Tranylcypromine 962
Triacylglycerols see “Glycerol esters”)
Tribromomethane see also “Bromoform”
Tribromomethane, dibromocarbene from 606—607
Tricarboxylic acid cycle 1123—1124
Trichloroacetic acid 801 802
Trichloromethane 166—167 (see also “Chloroform”)
Trichloromethane, boiling point of 150
Triethylamine 920
Trifluoroacetic acid 822
Trifluoroacetic acid, acidity of 41
Triglycerides see “Glycerol esters”
Trigonal planar geometry and 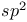 hybridization 89—91 157 191 430 706—707 hybridization 89—91 157 191 430 706—707
Trigonal planar geometry and VSEPR 30
Trigonal pyramidal geometry 30
Trimer 266
Trimethyl phosphate 641
Trimethyl phosphite 641
Trimethylamine 918
Trimethylamine H chemical shift 526
Trimyristin 853
Triose phosphate isomerase 1058
Tripeptide 1109
Triphenylamine 921
Triphenylmethane 621
Triphenylmethyl perchlorate 446
Triphenylphosphine 733
Triple bond 14 92—94 99 363 365—367
Triple bond in benzyne 982 984
Tristearin 846 1071—1072
Triterpenes 1085
Triterpenes, biosynthesis of 684—685 1089 1093—1095
Trityl see “Triphenylmethyl perchlorate”
Trivial names see “Common names”
Tropylium cation see “Cycloheptatrienyl cation”
Trypsin 1130
Twist-boat see “Skew boat conformation of cyclohexane”
Tyrian purple 4 54 974
Ubiquinone 1013
Ultraviolet-visible spectroscopy 565—567 577
Ultraviolet-visible spectroscopy alcohols 652
Ultraviolet-visible spectroscopy aldehydes and ketones 741
Ultraviolet-visible spectroscopy amines 953
Ultraviolet-visible spectroscopy carboxylic acids and derivatives 821 874
Ultraviolet-visible spectroscopy ethers and epoxides 691
Ultraviolet-visible spectroscopy phenols 1015
Unimolecular elementary step 156 159—160
Unimolecular elimination 206—208 217—219 223
Unimolecular nucleophilic substitution 160—161 180 339—345
Uracil 1157
Urea from ammonium cyanate 2
Urea, electrostatic potential map 1
Urea, industrial synthesis of 861
Urea, reaction of, with diethyl malonate 900
Urey, Harold C. 810
Uridine 1159
Uronic acids 1055
valence electrons 9
Valence electrons and Lewis structures 19—20
Valence-bond theory 60—61 92—93 95
Valence-shell electron pair repulsion and molecular geometry 29—31 49
van der Waals forces attractive 81—83
van der Waals forces attractive and stability of isomeric alkanes 87
van der Waals forces repulsive 82 109—110 117 121
van der Waals forces repulsive in stereoisomers 124—125 199—200 221
van der Waals radius 82 109
van der Waals strain 109 133
van der Waals strain 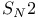 reactions 334—336 reactions 334—336
van der Waals strain alkenes 199—200 221
van der Waals strain axial substituents in cyclohexane 120—124 135
van der Waals strain boat conformation of cyclohexane 117
van der Waals strain butane 109 111 133—134
van der Waals strain in stereoisomers 124—125 199—200 221
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



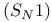
 of
of 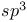 hybridization
hybridization unsaturated aldehydes and ketones
unsaturated aldehydes and ketones 
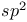 hybridization
hybridization 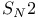 reactions
reactions