|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Fuhrhop J., Penzlin G. — Organic Synthesis: Concepts, Methods, Starting materials |

Обсудите книгу на
Нашли опечатку?
Выделите ее мышкой и нажмите Ctrl+Enter
|
Название: Organic Synthesis: Concepts, Methods, Starting materials
Авторы: Fuhrhop J., Penzlin G.
Язык: 
Рубрика: Химия/
Статус предметного указателя: Готов указатель с номерами страниц
ed2k:
Издание: second edition, rewise and enlarged
Год издания: 1994
Количество страниц: 432
Добавлена в каталог: 29.10.2005
Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
|
|
 |
| Предметный указатель |
Iodine = diiodine (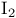 ), ),  isomerization of polyenes 31 isomerization of polyenes 31
Iodine = diiodine (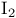 ), iodoetherification with 272 326 ), iodoetherification with 272 326
Iodine = diiodine (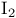 ), iodolactonization with 321 335—336 ), iodolactonization with 321 335—336
Iodine = diiodine (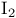 ), oxidative cleavage of organylzirconium 132 ), oxidative cleavage of organylzirconium 132
Iodine, phenylbis(trifluoroacetato)- + methanol (Stork reagent), 1,3-dithiane cleavage 328—329
Iodoalkanes see “Haloalkanes”
Iodoetherification 272 326
Iodolactones see “Halolactones”
Ion-exchange resins see “Benzene ethenyl-” “Sulfonic
Ionone,  - or - or  -, pr. 191 -, pr. 191
Ionone,  - or - or  -, synthesis 90—91 -, synthesis 90—91
Ionone, 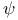 -, acid-catalysed cyclization of 90—91 -, acid-catalysed cyclization of 90—91
Ipc (isopinocampheyl), chiral auxiliary group 108
Ireland — Claisen rearrangement 87 325
Iron(1+),  -, addition of carbon nucleophiles 44 -, addition of carbon nucleophiles 44
Iron(1+),  -, addition of carbon nucleophiles 44 -, addition of carbon nucleophiles 44
Iron, 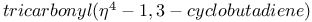 - 78 - 78
Iron,  -, coupling with alkenes 44 -, coupling with alkenes 44
Iron,  -, ox. coupling with carbon nucleophiles 44 -, ox. coupling with carbon nucleophiles 44
Iron, anionic carbonyl complexes see “Ferrate ...”
Isobutyryl chloride,  -acetoxy- 160 -acetoxy- 160
Isocyanates, blocking of OH with 301
Isocyanates, blocking of SH with 229 238
Isocyanates, synthesis 143 301 331
Isocyanides, trapping of diallylnickel 41
Isodrin, pagodane and dodecahedrane from 336—337
Isodrin, pr. 192
Isoephedrine, 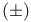 - 302 - 302
Isomerization, configurational,  and/or and/or  of alkenes via 1,2-diol thiocarbonates 142 of alkenes via 1,2-diol thiocarbonates 142
Isomerization, configurational,  and/or and/or  of alkenes with hydrogenation catalyst 100—102 of alkenes with hydrogenation catalyst 100—102
Isomerization, configurational,  and/or and/or  of alkenes, photochemical 31 282 296 332 of alkenes, photochemical 31 282 296 332
Isomerization, configurational,  and/or and/or  of alkenes, thermal or iodine-catalysed 31 of alkenes, thermal or iodine-catalysed 31
Isomerization, configurational, cis/trans or 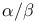 at rings see “Epimerization” at rings see “Epimerization”
Isomerization, constitutional see “Rearrangements”
Isopinocampheyl, chiral auxiliary group 108
Isoprene see “1 2-methyl-”
Isopropamide iodide, 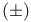 - 302 - 302
Isoquinolines 292—293
Isoquinuclidine derivs. = 2-azabicyclo[2.2.2]-octane derivs., retro-synthesis 212—213
Isoquinuclidine derivs. = 2-azabicyclo[2.2.2]-octane derivs., synthesis 153 291 297
Isothioureas, S-alkyl-, thiols from 168
Isoureas, O-acyl- = carbamimidic anhydrides, reactive intermediates 144—145 234
Isoureas, O-alkyl- = carbamimidic esters 144—145
Isoxazoles 307—308
Isoxazoles, 4,5-dihydro- 153
Isoxazolidines 153
Jasmone = (Z)-3-methyl-2-(2-pentenyl)-2-cyclopenten-1-one 29
Jones’ reagent see “Chromium(6+) oxide with sulfuric acid”
Juglone = 5-hydroxy-1,4-naphthalenedione, dienophilc in tetracycline synthesis 318
Julia — Lythgoe olefination 34
Julia’s terpenoid synthesis 69—70
Juvenile hormone, synth. steps 20 30 135 155
KAPA see “1 mono-K
Katsuki — Sharpless oxn. see “Sharpless epoxidation”
Kekulene = 15,23:16,22-dimethenobenzo[1,2-a:5,4-a']dipentaphene 338
Kemp’s acid 346—347
Kemp’s acid, molecular complexes of derivatives 346—348
Ketenes see “Ethenone”
Ketimines see “Imines”
Ketones,  -(alkylsulflnyl)-, synthesis and conversions 50—51 65 -(alkylsulflnyl)-, synthesis and conversions 50—51 65
Ketones,  -acidity of 10 -acidity of 10
Ketones,  -activation of see “Enamines” “Enolates” “Vinylogous -activation of see “Enamines” “Enolates” “Vinylogous
Ketones,  -acylation of 81 -acylation of 81
Ketones,  -acylation of, via enamines 88 -acylation of, via enamines 88
Ketones,  -acylation of, via sulfur extrusion 59—60 -acylation of, via sulfur extrusion 59—60
Ketones,  -acylthic, S extrusion from 59—60 -acylthic, S extrusion from 59—60
Ketones,  -alkoxy-, by oxn. of alkynes 117 132 -alkoxy-, by oxn. of alkynes 117 132
Ketones,  -alkylation of 24—26 82 87 -alkylation of 24—26 82 87
Ketones,  -amino-, pyrroles from 150—151 -amino-, pyrroles from 150—151
Ketones,  -amino-, synthesis from enol ethers 268 -amino-, synthesis from enol ethers 268
Ketones,  -amino-, synthesis from ketones 147 151 302 -amino-, synthesis from ketones 147 151 302
Ketones,  -halo-, -halo-, 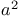 -synthons 2 16 18 -synthons 2 16 18
Ketones,  -halo-, -halo-,  -alkylation with C nucleophiles 63 -alkylation with C nucleophiles 63
Ketones,  -halo-, -halo-,  -amination 302 -amination 302
Ketones,  -halo-, Favorskii rearr. of 78 84 -halo-, Favorskii rearr. of 78 84
Ketones,  -halo-, reduction to oxiranes, enantioselective 108 -halo-, reduction to oxiranes, enantioselective 108
Ketones,  -hydroxy- (acyloins), Birch type a-deoxygenation 156 277 -hydroxy- (acyloins), Birch type a-deoxygenation 156 277
Ketones,  -hydroxy- (acyloins), conversion to imidazoles 151—152 -hydroxy- (acyloins), conversion to imidazoles 151—152
Ketones,  -hydroxy- (acyloins), synthesis of, by oxidation of alkynes 132 -hydroxy- (acyloins), synthesis of, by oxidation of alkynes 132
Ketones,  -hydroxy- (acyloins), synthesis of, by oxidation of ketones 116 121—122 -hydroxy- (acyloins), synthesis of, by oxidation of ketones 116 121—122
Ketones,  -hydroxy- (acyloins), synthesis of, by redn. of 1,2-diones with -hydroxy- (acyloins), synthesis of, by redn. of 1,2-diones with  219 219
Ketones,  -hydroxy- (acyloins), synthesis of, from carbonyl compounds with 1,3-dithiane anions 51 -hydroxy- (acyloins), synthesis of, from carbonyl compounds with 1,3-dithiane anions 51
Ketones,  -hydroxy- (acyloins), synthesis of, from carbonyl compounds with 1-alkyne anions 52 -hydroxy- (acyloins), synthesis of, from carbonyl compounds with 1-alkyne anions 52
Ketones,  -hydroxy- (acyloins), synthesis of, from carboxylic acid esters by acyloin coupling 53—54 -hydroxy- (acyloins), synthesis of, from carboxylic acid esters by acyloin coupling 53—54
Ketones,  -hydroxy- (acyloins), synthesis of, from carboxylic acid esters with DMSO anion 51 -hydroxy- (acyloins), synthesis of, from carboxylic acid esters with DMSO anion 51
Ketones,  -dehydrogenation of 138—140 -dehydrogenation of 138—140
Ketones,  -unsaturated, -unsaturated,  - and - and 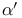 -alkylation of 24—25 82 277 -alkylation of 24—25 82 277
Ketones,  -unsaturated, -unsaturated,  , p-dialkylation 277 , p-dialkylation 277
Ketones,  -unsaturated, -unsaturated,  -alkylation of 20—21 277 -alkylation of 20—21 277
Ketones,  -unsaturated, conversion to cyclobutanones 75/77 -unsaturated, conversion to cyclobutanones 75/77
Ketones,  -unsaturated, conversion to cyclopentanones 75/77 83 -unsaturated, conversion to cyclopentanones 75/77 83
Ketones,  -unsaturated, Favorskii rearr. of 84 211 -unsaturated, Favorskii rearr. of 84 211
Ketones,  -unsaturated, hydration of 274—275 -unsaturated, hydration of 274—275
Ketones,  -unsaturated, oxidative cleavage of 82 -unsaturated, oxidative cleavage of 82
Ketones,  -unsaturated, reduction of, to alkenes 109 -unsaturated, reduction of, to alkenes 109
Ketones,  -unsaturated, reduction of, to allylic alcohols 105—107 -unsaturated, reduction of, to allylic alcohols 105—107
Ketones,  -unsaturated, reduction of, to enolates with Li 73 -unsaturated, reduction of, to enolates with Li 73
Ketones,  -unsaturated, reduction of, to enolates with RMgBr 281 -unsaturated, reduction of, to enolates with RMgBr 281
Ketones,  -unsaturated, reduction of, to ketones with -unsaturated, reduction of, to ketones with 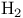 /catalyst 283 /catalyst 283
Ketones,  -unsaturated, reduction of, to ketones with Li 73 103—104 -unsaturated, reduction of, to ketones with Li 73 103—104
Ketones,  -unsaturated, synthesis of, by -unsaturated, synthesis of, by  -dehydrogenation of ketones 122 138—140 -dehydrogenation of ketones 122 138—140
Ketones,  -unsaturated, synthesis of, by -unsaturated, synthesis of, by  -eliminations 57 138 140 274 -eliminations 57 138 140 274
Ketones,  -unsaturated, synthesis of, by -unsaturated, synthesis of, by  -eliminations of sulfoxides 65 86 -eliminations of sulfoxides 65 86
Ketones,  -unsaturated, synthesis of, by aldol type condensations 59 71—73 81 82 280 -unsaturated, synthesis of, by aldol type condensations 59 71—73 81 82 280
Ketones,  -unsaturated, synthesis of, by Horner oleflnation 29 -unsaturated, synthesis of, by Horner oleflnation 29
Ketones,  -unsaturated, synthesis of, by Mannich reactions 57 -unsaturated, synthesis of, by Mannich reactions 57
Ketones,  -unsaturated, synthesis of, by oxidation of allylic -unsaturated, synthesis of, by oxidation of allylic  116 119—120 116 119—120
Ketones,  -unsaturated, synthesis of, by oxidation of allylic alcohols 118 133—135 323 -unsaturated, synthesis of, by oxidation of allylic alcohols 118 133—135 323
Ketones,  -unsaturated, synthesis of, by reduction of alkoxyarenes 278 -unsaturated, synthesis of, by reduction of alkoxyarenes 278
Ketones,  -amino- (Mannich bases), -amino- (Mannich bases),  -elimination of 57 72—73 -elimination of 57 72—73
Ketones,  -amino- (Mannich bases), pr. 178 -amino- (Mannich bases), pr. 178
Ketones,  -amino- (Mannich bases), synthesis 57 300 304 -amino- (Mannich bases), synthesis 57 300 304
Ketones,  -hydroxy-, synthesis by aldol type addn 55—58 60—62 -hydroxy-, synthesis by aldol type addn 55—58 60—62
Ketones,  -hydroxy-, synthesis by hydration of 2-enones 274—275 -hydroxy-, synthesis by hydration of 2-enones 274—275
Ketones,  -unsaturated, synth. by Torgov reaction 71 278—279 -unsaturated, synth. by Torgov reaction 71 278—279
Ketones, (triphenylphosphoranylidene)hydrazones, Barton olefination of thiones with 35
Ketones, 1,2-oxygen shift procedure 156
Ketones, 1-alkenylation of 62—63
Ketones, 1-alkylation of 44
Ketones, 1-alkynylation of 52 62—63 278
Ketones, 1-allylation of 66—69
Ketones, conversion of, to  -chloro aldehydes ( -chloro aldehydes ( ) 51—52 ) 51—52
Ketones, conversion of, to  -hydroxy aldehydes (TosMIC) 49 51 -hydroxy aldehydes (TosMIC) 49 51
Ketones, conversion of, to aldehydes ( ) 48—49 ) 48—49
Ketones, conversion of, to cyclopropanol silyl ethers 75 83
Ketones, conversion of, to enamines 13 88
Ketones, conversion of, to enol silyl ethers 11 58 75 83
Ketones, conversion of, to enolates 11—12 24—25 58
Ketones, conversion of, to nitriles (TosMIC) 49
Ketones, conversion of, to oxiranes (sulfonium ylides) 28 45
Ketones, cyclic, 5-membered, synthesis 81—83
Ketones, cyclic, selective monoalkylation 25—26
Ketones, cyclic, synthesis, Birch redn. of alkoxyarenes 103—104 278
Ketones, cyclic, synthesis, from dienes and  47—48 47—48
Ketones, homologization of, by diazoalkanes 83 166
Ketones, olefination 28—35 41 110 267
Ketones, oxidative a-coupling via enol ethers 65
Ketones, oxidative cleavage of, Beckmann rearr. to lactams 136—137
Ketones, oxidative cleavage of, by  via enol ethers 285 via enol ethers 285
Ketones, oxidative cleavage of, by  via via  -(arylmethylene) ketones 82 -(arylmethylene) ketones 82
Ketones, oxidative cleavage of, by 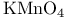 137 137
Ketones, oxidative cleavage of, by fragmentation via a-dithioketals 79
Ketones, oxidative cleavage of, to esters or lactones 80 136 165 206 210 276 282 283 319—320
Ketones, protection of see “Protection of carbonyl groups”
Ketones, rearrangements via tosylhydrazones 141—142
Ketones, reduction of, to alcohols 98 105—108
Ketones, reduction of, to alcohols, enantiosel 102—103 108 325—326
Ketones, reduction of, to alkanes 98 109
Ketones, reduction of, to alkenes 141—142
Ketones, reduction of, to amines via imines or oximes 98 112
| Ketones, reductive coupling of 35 41 53
Ketones, reductive coupling of, with 2-enoic esters 69
Ketones, synthesis of, form aldehydes via 1,3-dithianes 22 51
Ketones, synthesis of, form aldehydes via dicyanomethylene derivs. 166
Ketones, synthesis of, form alkenes via boranes + CO 47—48
Ketones, synthesis of, form alkenes, ox. cleavage 87—88 156—157 280 282
Ketones, synthesis of, form alkenes, ox. rearr. with  129—130 129—130
Ketones, synthesis of, from 5(4H)-oxazolones + 2RX 48
Ketones, synthesis of, from arf-nitroalkanes, aq. acid 16 65—66
Ketones, synthesis of, from carboxylic acid derivs.,  46—47 46—47
Ketones, synthesis of, from carboxylic acid derivs., halides, 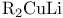 or RZnX 45—46 or RZnX 45—46
Ketones, synthesis of, from carboxylic acid derivs., lithium salts, RLi 45—46 282
Ketones, synthesis of, from carboxylic acid derivs., nitriles, RMgX 201 298—299
Ketones, synthesis of, from carboxylic acid derivs., thioesters, RLi or RMgX 319—320
Ketones, synthesis of, from diallylic Ni complexes + RNC 41
Ketones, synthesis of, from dithioacetals + RX 22 51 66 205
Ketones, synthesis of, from enol or enamine derivatives, hydrolysis 48—49 103—104 278
Ketones, synthesis of, from enol or enamine derivatives, ox. cleavage with  or or 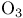 280 285 280 285
Ketones, synthesis of, from hydrazones, 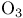 26 26
Ketones, synthesis of, from nitroalkenes, reduction 301
Ketones, synthesis of, from sec. alcohols by oxidation 133—135 267 276—277 283 322—323 328—329 337
Ketones, synthesis of, from tetracarbonylferrate(2-)/CO 46—47
Ketones, synthesis of, from thioenol ethers by hydrolysis 83
Kinetic control, diastereotoposelective lactonization 168
Kinetic control, directed aldol addns. 56—62
Kinetic control, enolate formation 11—13 24—25 58
Kinetic control, Wittig olefination 28—31
Kinetic protonation of enolates 322—323
Kinetic resolution see “Optical resolution”
Knoevenagel condensation 56 58—59 166
Knorr’s pyrrole synthesis 150—151
Koenigs — Knorr glycosylation with glycosyl halides and mercury(2+) 267
L-Arabinitol, 1,2:4,5-dianhydro-, prepn. 160 327—328
L-Arabinitol, chiral educt 160 327—328
L-Arginine, pr. 184
L-Arginine, protection 229
L-Cysteine, cephalosporin from 313—314
L-Cysteine, pr. 185
L-Cysteine, S-protection 229
L-Glutamic acid, chiral educt 202—203
L-Glutamic acid, pr. 184
L-Glutamic acid, protection 229
L-Hexoses, total synthesis 264—265
L-Homoproline, FK 506 synthon 324 326—327
L-Mandelic acid = (S)-hydroxyphenylacetic acid, chiral auxiliary from 62
L-Proline, chiral ligand from 20—21
L-Proline, N,O-protection 299
L-Proline, stereoselectivity  -alkylation 299 -alkylation 299
L-Serine,  -lactams from serine amides 161 -lactams from serine amides 161
L-Serine, educt for chiral auxiliary 23
L-Serine, pr. 184
L-Serine, protection 229
L-Streptose, synthesis 267
L-Valinol = (S)-2-amino-3-methyl-butanol, chiral auxiliary 61
Lactams, macrocyclic, synthesis 240—241 247 249—250 328—329
Lactams, pr. 178 184
Lactams, synth. from cyclic ketoximes 136—137 195
Lactones,  -alkylation, diastereoselective 321 322 327—328 -alkylation, diastereoselective 321 322 327—328
Lactones, macrocyclic, synthesis by intramol. allyl coupling 42
Lactones, macrocyclic, synthesis by intramol. esterification 145—146
Lactones, reduction to cyclic ethers 110—111
Lactones, reduction to cyclic hemiacetals 105 321 322 327—328
Lactones, synthesis by Baeyer — Villiger oxidation 80 136 165 206 210 276 319—320
Lactones, synthesis by halolactonization 275 319—320 321
Lactones, synthesis by intramol. esterification 63—65 69 70 145—146 168 273 322 325 327—328
Ladenburg benzene = prismane 330—331
Lanosta-8,24-dien-3-ol,  - (lanosterol), biosynthesis, syntheses relating to 279—280 - (lanosterol), biosynthesis, syntheses relating to 279—280
Lanosta-8,24-dien-3-ol,  - (lanosterol), pr. 284 - (lanosterol), pr. 284
Lawesson type reagents 110—111
LDA see “2-Propaneamine N-(1-methylethyl)- lithium
Lead(2+) acetate, additive for Lindlar cat 100
Lead(2+) acetate, basic acetate,  , catalyst for aldol type condensation 318 , catalyst for aldol type condensation 318
Lead(4+) acetate, oxidation with, of alcohols to aldehydes 117 133
Lead(4+) acetate, oxidation with, of amines to nitrenes 212—213
Lead(4+) acetate, oxidation with, of hydrazines to diazenes 35
Lead(4+) acetate, oxidative degradation of 1,2-dicarboxylic acids 80 142 333
Lead(4+) acetate, oxidative degradation of a-amino acids 48
Lead(4+) acetate, oxidative degradation of carboxylic acids with  337 337
Lecithins, vesicle membranes 350
Leukotriene 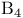 , synthesis 125—126 , synthesis 125—126
Lewis acids see “Aluminum(3+) chloride” “Borane trifluoro-” “Lithium(1+)” “Magnesium(2+)” “Mercury(2+)” “Silvei(1+)” “Thallium(1+)” “Tin(4+) “Titanium(4+) “Zinc(2+)”
Librium 307
Ligands, macrocyclic 235—237 241 247—262
Lindlar catalyst 100
Lindlar catalyst, hydrogenation of alkynes 40 64 100—101
Linear synthesis vs. convergent synthesis, disadvantages 224—225 233—234
Lipase, pig pancreatic 167
Lipid membranes 350—351
Lithium(1+), complex or organic salts (except organyls) see corresponding acid or anion
Lithium(1+), perchlorate, cat. for Diels — Alder addition 86
Lithium(1+), salts, cat. for dehydrogenation 52 140
Lithium(1+), salts, cat. for rearr. of oxiranes 52 77 79
Lithium, (1,1-dimethylethyl)- ( ), halogen-metal exchange with 70 326—327 ), halogen-metal exchange with 70 326—327
Lithium, (1,1-dimethylethyl)- ( ), lithiation with, of sulfones 8 ), lithiation with, of sulfones 8
Lithium, (1,1-dimethylethyl)- ( ), lithiation with, of thioesters 323 ), lithiation with, of thioesters 323
Lithium, (1,1-dimethylethyl)- ( ), prepn. of ), prepn. of 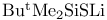 from 169 from 169
Lithium, (1,3-dithian-2-yl)- see “1
Lithium, (1-methylpropyl)- ( ), lithiation of allylic CH groups 14 15 ), lithiation of allylic CH groups 14 15
Lithium, (2-methoxycyclopropyl)-, cis-, 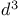 -synthon 14—15 19 70 76 -synthon 14—15 19 70 76
Lithium, (2-methoxycyclopropyl)-, cis-, prepn. 70
Lithium, (ethoxyethynyl)-, carboxymethyl 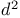 -synthon 64—65 325 327—328 -synthon 64—65 325 327—328
Lithium, (triphenylmethyl)- (trityllithium,  ), selective enolization with 11—12 ), selective enolization with 11—12
Lithium, alkyl-, addn. to 1-aikenyl silanes 34
Lithium, alkyl-, addn. to lithium carboxylates 45—46 283
Lithium, alkyl-, addn. to thionolactones 110—111
Lithium, butyl- (BuLi) 5
Lithium, butyl- (BuLi), lithiation with, of 1-alkynes 64—65 303 325 328
Lithium, butyl- (BuLi), lithiation with, of 1-alkynes, 1,3-dilithiation 281
Lithium, butyl- (BuLi), lithiation with, of allylic and benzylic CH groups 10 14
Lithium, butyl- (BuLi), lithiation with, of carboxylic acids (dilithiation) 63
Lithium, butyl- (BuLi), lithiation with, of dithioacetals 6 8 22 51 66
Lithium, butyl- (BuLi), lithiation with, of imidic esters 22—23
Lithium, butyl- (BuLi), lithiation with, of phosphonic diamides 328 329
Lithium, butyl- (BuLi), lithiation with, of phosphonium salts 6 31—32
Lithium, butyl- (BuLi), lithiation with, of sec. carboxamides 61
Lithium, butyl- (BuLi), lithiation with, of silanes (activated) 7
Lithium, butyl- (BuLi), lithiation with, of sulfones 34
Lithium, butyl- (BuLi), lithiation with, of sulfonium salts 45 154
Lithium, butyl- (BuLi), lithiation with, of tosylhydrazones 141
Lithium, dichloromethyl, addn. to ketones 51—52
Lithium, ethenyl- (vinyllithium) 5
Lithium, ethynyl-, addition to oxiranes 321
Lithium, metalation of organyl halides with 5—6
Lithium, methyl-, alkyl d-synthon 1—2 79 283
Lithium, methyl-, cleavage of silyl enolates with 57—58 73
Lithium, methyl-, halogen-metal exchange with 74—75
Lithium, methyl-, lithiation with, of sulfoxides 9
Lithium, methyl-, lithiation with, of tosylhydrazones 141
Lithium, methyl-, metal-metal exchange, with Cu(1+) 5 20
Lithium, methyl-, transmetalation of organylstannanes 322—323
Lithium, reduction with see “Birch reduction”
Lithium, reductive desulfurization of sulfones 70
Lithium, [(methylthioXtrimethylsilyl)methyl]-, Peterson olefination with 34
Lithium-biphenyl reagent, 10-demethylation of steroids 287—288
Lithium-naphthalene reagent, deprotonation of 1-alkynes 52
Lysine, pr. 184
Lysine, protection 229 241
Macrocycles see “Carbocycles macrocyclic”
Macrocycles, lactams 247 249—250 324—329
Macrocycles, lactones 42 145—146 319—329
Macrocycles, ligands 235—236 247 249—250
Macrocycles, peptides 235—236 241
Macrocycles, peptides, solid-phase method 241
Macrolides see “Lactones macrocyclic”
Magnesium(2+) halides, catalysts, for allylstannane addn. to RR'CO 66—67
Magnesium(2+) halides, catalysts, for cyclopropanemethanol opening 70 77
Magnesium(2+) halides, transmetalation of organyllithium 326—327
Magnesium, alkylhalo-, metal-metal exchange of 5 7 20 46
Magnesium, alkylhalo-, preparation 7 20 46 326—327
Magnesium, bromoethenyl-, ethenylation with 18 62—63
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



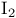 ),
),  isomerization of polyenes
isomerization of polyenes  - or
- or  -, pr.
-, pr.  -, acid-catalysed cyclization of
-, acid-catalysed cyclization of  -, addition of carbon nucleophiles
-, addition of carbon nucleophiles  -, addition of carbon nucleophiles
-, addition of carbon nucleophiles 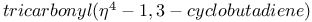 -
-  -, coupling with alkenes
-, coupling with alkenes  -
-  and/or
and/or 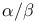 at rings
at rings 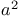 -synthons
-synthons 
 -dehydrogenation of
-dehydrogenation of 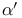 -alkylation of
-alkylation of 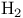 /catalyst
/catalyst 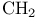
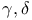 -unsaturated, synth. by Torgov reaction
-unsaturated, synth. by Torgov reaction 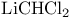 )
)  )
) 
 via enol ethers
via enol ethers 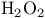 via
via 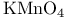


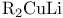 or RZnX
or RZnX 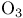
 - (lanosterol), biosynthesis, syntheses relating to
- (lanosterol), biosynthesis, syntheses relating to  , catalyst for aldol type condensation
, catalyst for aldol type condensation 
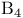 , synthesis
, synthesis  ), halogen-metal exchange with
), halogen-metal exchange with 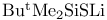 from
from  ), lithiation of allylic CH groups
), lithiation of allylic CH groups 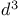 -synthon
-synthon 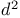 -synthon
-synthon  ), selective enolization with
), selective enolization with