|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Fuhrhop J., Penzlin G. — Organic Synthesis: Concepts, Methods, Starting materials |

Обсудите книгу на
Нашли опечатку?
Выделите ее мышкой и нажмите Ctrl+Enter
|
Название: Organic Synthesis: Concepts, Methods, Starting materials
Авторы: Fuhrhop J., Penzlin G.
Язык: 
Рубрика: Химия/
Статус предметного указателя: Готов указатель с номерами страниц
ed2k:
Издание: second edition, rewise and enlarged
Год издания: 1994
Количество страниц: 432
Добавлена в каталог: 29.10.2005
Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
|
|
 |
| Предметный указатель |
Ferrate(2-), tetracarbonyl-, formyl d-synthon 18 46—47 198
Ferrate(2-), tetracarbonyl-, redn. of acid anhydrides and halides with 46—47
Ferrate(3-), hexakis(cyano-C)-, potassium ( ), ), 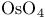 -catalysed oxn. with 129 -catalysed oxn. with 129
FGA, retro-synthetic 195
FGI 95
FGI, retro-synthetic 195
FGR, retro-synthetic 195
Fischer formula, erythro/threo definition 62 360
Fischer’s indole synthesis 151—152 296 307
Fischer’s porphyrin synthesis 256
FK 506, total synthesis 324—329
Fluoride, hydrogen, cleavage with, of diol acetonides 277
Fluoride, hydrogen, cleavage with, of silyl ethers 277 328 329
Fluoride, hydrogen, cleavage with, of silyl thioethers 169
Fluoride, potassium, base cat. for Michael addn. 71
Fluoride, silver, halide/fluoride exchange 269—270
Fluoride, tetrabutylammonium, cleavage of silyl ethers 157 159 270 272 281 339
Fluorination with 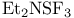 (DAST), OH/F exchange 269—270 272 (DAST), OH/F exchange 269—270 272
Fluorination with 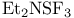 (DAST), SR/F exchange ( (DAST), SR/F exchange (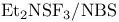 ) 269—270 ) 269—270
Fluorination with  , oxidation of alkanes 118—119 157 , oxidation of alkanes 118—119 157
Fluorosulfuric acid, methyl ester 338
Formaldehyde, hydroxymethyl 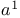 -synthon 18 198 203 302 -synthon 18 198 203 302
Formaldehyde, Mannich reactions 57 292 300 304 310 317—318
Formaldehyde, reduction of aldehydes with 302
Formaldehyde-sulfoxylate see “Methanesulfinic acid hydroxy-”
Formamides, N,N-diaikyl-, formyl 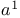 -synthons 16 18 198 255 -synthons 16 18 198 255
Formic acid, azido- see “Carbonazidic acid”
Formic acid, chloro- see “Carbonochloridic acid”
Formic acid, esters, formyl 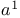 -synthons 18 58—59 155—156 337 -synthons 18 58—59 155—156 337
Formic acid, esters, orthoesters see “Orthoformic acid esters”
Formylation, 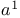 -synthons 16 18 -synthons 16 18
Formylation, 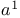 -synthons, formic esters 59 155—156 -synthons, formic esters 59 155—156
Formylation, 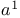 -synthons, orthoformamidic esters 79 82 -synthons, orthoformamidic esters 79 82
Formylation, 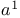 -synthons, orthoformic esters/trifluoroborane 59 -synthons, orthoformic esters/trifluoroborane 59
Formylation, 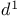 -synthons 6—8 18 -synthons 6—8 18
Formylation, 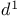 -synthons, (dichloromethyl)lithium 51—52 -synthons, (dichloromethyl)lithium 51—52
Formylation, 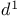 -synthons, 1,3-dithianes 17—18 198 267 -synthons, 1,3-dithianes 17—18 198 267
Formylation, 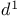 -synthons, allyl or vinyl d-synthons + -synthons, allyl or vinyl d-synthons + 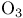 90 90
Formylation, 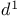 -synthons, boranes + carbon monoxide 47—48 -synthons, boranes + carbon monoxide 47—48
Formylation, 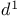 -synthons, cyanide anion 50 -synthons, cyanide anion 50
Formylation, 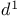 -synthons, DMSO anion 51 -synthons, DMSO anion 51
Formylation, 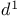 -synthons, isocyanomethyl sulfones (TosMIC) 49 51 -synthons, isocyanomethyl sulfones (TosMIC) 49 51
Formylation, 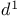 -synthons, nitromethane anion 65—66 -synthons, nitromethane anion 65—66
Formylation, 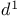 -synthons, tetracarbonylferrate(2-) 46—47 -synthons, tetracarbonylferrate(2-) 46—47
Formylation, reductive, (alkoxymethylene)phosphoranes 6 49
Fragmentation reactions 89—90 141—142
Frater — Seebach alkylation 27 325—326
Friedel — Crafts acylation, 1-oxoalkylium (acylium) ions in 15—16
Friedel — Crafts acylation, aldol type cyclizations by 81
Friedel — Crafts alkylation of dipyrromethenes 256—257
Friedel — Crafts alkylation of indoles 291
Friedel — Crafts alkylation of polystyrene 233
Friedel — Crafts alkylation, alkylium ions in 15—16
Fullerenes 357
Fullerenes, metal inclusion compounds 357
Fulvalene see “Bi-2
Functional group addition (FGA), rerro-synthetic 195
Functional group interconversion (FGI) 95—169
Functional group interconversion (FGI), retro-synthetic 195
Functional group removal (FGR), retro-synthetic 195
Functional group, definition 1—4 50
Furan, Diels — Alder addition to 86 93
Furosemide 305
Gene cloning 241—246
Genetic engineering methods 225—227 241—246
Geraniol see “2 3
Geranyl bromide see “2 1-bromo-3
Gipsy moth pheromone 34 125
Girgnard tranform, retro-synthetic 194
Glaser coupling 40
Glass, porous, solid support 221 342
Glucal derivatives 268
Glucosamine, pr. 263
Glucosamine, synthesis 268
Glucose derivatives, pr. 263—264
Glucose derivatives, syntheses of 266—269
Glucose derivatives, syntheses starting from 267—273
Glutethimide, 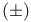 - 304 - 304
Glycals 268
Glycine, N-benzoyl,  synthon 48 synthon 48
Glycols see “Diols”
Glycosyl fluorides 269—270 272
Glycosyl halides,  -elimination, 2-acetoxyglycals 268 -elimination, 2-acetoxyglycals 268
Glycosyl halides, glycosylations with 267 269—271
Glycosyl halides, prepn. 266 269—270 272
Glycosyl halides, reductive  -elimination, glycals 268 -elimination, glycals 268
Glycosyl imidates 270—271
Glycosylations 267 269—272
Glyoxylic acid = oxoacetic acid, derivs., deblocking of acetals with 328—329
Glyoxylic acid = oxoacetic acid, derivs., pr. 176
Glyoxylic acid = oxoacetic acid, derivs., synthesis 202
Grignard reagents see “Magnesium alkylhalo-”
Griseofulvin, 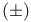 -, synthesis 74 -, synthesis 74
Griseofulvin, dehydro-, 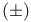 -, synthesis 86 -, synthesis 86
Guanidine group, protection in arginine 229
Hafnium,  -, + -, + 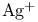 , activation of glycosyl fluorides 270 , activation of glycosyl fluorides 270
Haloalkanes, alkenes from see “ -Elimination” -Elimination”
Haloalkanes, alkyl a-synthons 15—16 22—27
Haloalkanes, conversion to phosphines 282
Haloalkanes, conversion to thiols 169
Haloalkanes, coupling of 19—20 36
Haloalkanes, metalation (= umpolung) 5—7 17 20 70 75 326—327
Haloalkanes, reduction of 97 113—115 203
Haloalkanes, reduction of, with alkali metals/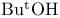 (Birch redn.) 337 (Birch redn.) 337
Haloalkanes, reduction of, with chromium(2 +) acetate 286
Haloalkanes, reduction of, with hydrogen/Raney Ni 287
Haloalkanes, reduction of, with methyllithium/lithium bromide 75
Haloalkanes, reduction of, with tributylstannane 97 114—115 275 319—320
Haloalkanes, reduction of, with zinc or zinc-copper 83 335—336
Haloalkanes, synthesis from alcohols, bromination, conc. aq.  303 303
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/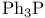 169 169
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/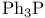 , fluorination with , fluorination with 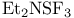 269—270 272 269—270 272
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/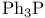 , glycosyl halides 266 269—270 272 , glycosyl halides 266 269—270 272
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/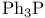 , iodides via tosylates 203 281 , iodides via tosylates 203 281
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/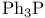 , iodides, iodine/ , iodides, iodine/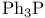 328 328
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/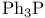 , Mitsunobu reactions with , Mitsunobu reactions with  160—161 160—161
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/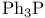 , tetrahalomethane/ , tetrahalomethane/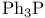 266 355 266 355
Haloalkanes, synthesis from alcohols, chlorination, N-Chlorosuccimmide/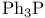 , thionyl chloride ( , thionyl chloride (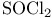 ) 32 ) 32
Haloalkanes, synthesis from alcohols, chlorination, N-chlorosuccinimide 282
Haloalkanes, synthesis from alcohols, chlorination, synthesis from thioethers 269—270
Halohydrins (vic-halo alcohols), synthesis of, from 1,2-diols 160 327—328
Halohydrins (vic-halo alcohols), synthesis of, from alkenes 77 117 275—276 287
Halohydrins (vic-halo alcohols), synthesis of, from oxiranes, with HF 287
Halolactonization 275 319—321 335—336
Hauser’s rule 9 24 204
HBT = 1-hydroxy-1H-benzotriazole 145 231
Heck coupling (alkene + organyl halide) 42—43
Hectane, synthesis 36
Helicate complexes 345—346
Hemiacetals, cyclic, from lactones ( ) 105 321 322 327—328 ) 105 321 322 327—328
Heterocycles see “Alkaloids” “Drugs” “N-Heterocycles”
Heterocycles, 3-membered see “Aziridines” “Oxiranes” “Thiiranes”
Heterocycles, macrocyclic see “Macrocycles”
Hexamethyldisilazane see “Stlanamine ...”
Hexanoic acid, 2-methyl-, (S)-, synth. 22—23
Hexonamides, N-alkyl-, aqueous suspensions, various micellar aggregates 352—353
High-dilution methods, macrocycle syntheses by 145—146 240—241 329 338
Hindrance, steric see “Steric hindrance”
Hippuric acid see “Glycine N-benzoyl-”
HMDS see “Silanamine ...”
HMPA = HMPTA see “Phosphoric triamide hexamethyl-”
HOBT = 1-hydroxy-1H-benzotriazole 145 231
Hofmann degradation 139 308 331
Hofmann type elimination of ammonium compounds 57 72 138 140—141 331
Hofmann type elimination of sulfonium compounds 38 338
Hofmann type elimination of sulfoxides 65 86 315
Homo-DNA 344—345
homo-Michael-type additions,  69—70 69—70
homo-Michael-type additions, 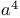 + alkenyl d 276 + alkenyl d 276
Homoenolate synthons 14—15 70 76
| Horner olefination 28—30 (see also “Phosphonic acids alkyl- diesters”)
Huenig base see “1-Naphthalenamine N
Huenig bases, polymeric = aminated chloromethylated ethenylbenzene homopolymers, deprotonation with, of ketones 11
Huenig bases, polymeric = aminated chloromethylated ethenylbenzene homopolymers, deprotonation with, or phosphonium salts 32
Huenig bases, polymeric = aminated chloromethylated ethenylbenzene homopolymers, removal of acids with 32
Hybrid plasmids 243—245
hydration see “Alkenes” “Alkynes”
Hydrazine, hydrazinolysis with, of esters 239—240 331
Hydrazine, hydrazinolysis with, of phthaloyl-protected amines 162 163
Hydrazine, reduction of ketones with 97—98 109
Hydrazines, cyclic, oxn. 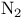 extrusion 35 331 extrusion 35 331
Hydrazones, aryl-, indole synth. with 151—152 296 307
Hydrazones, dialkyl-, lithiated, alkylation 12 18 25—26
Hydrazones, dialkyl-, lithiated, alkylation, enantioselective (SAMP-hydrazones) 25—26
Hydrazones, dialkyl-, lithiated, ozonolysis of 26
Hydrazones, Wolff — Kishner redn. of 109
Hydrazones, [(4-methylphenyi)sulfonyl]- (tosylhydrazones), fragmentation of 89 141—142
Hydrazones, [(4-methylphenyi)sulfonyl]- (tosylhydrazones), reduction of, to alkanes 109
Hydrazones, [(4-methylphenyi)sulfonyl]- (tosylhydrazones), reduction of, to alkenes 141—142
Hydrides of Al, B, Sn see “Aluminum” “Aluminate” “Borane” “Borate” “Stannane”
Hydrides, alkali, deprotonation with, of alcohols 23 33 157 325 328
Hydrides, alkali, deprotonation with, of carbonyl compounds 15 49 58 63 93
Hydrides, alkali, deprotonation with, of carboxamides 314
Hydrides, alkali, deprotonation with, of indoles 307
Hydrides, alkali, deprotonation with, of phosphonic diesters 29 334
Hydrides, alkali, deprotonation with, of sulfonium salts 38 45
Hydrides, alkali, deprotonation with, of sulfoxides 6 51
Hydrides, alkali, deprotonation with, of tosylhydrazones 35
Hydroalkylation of alkenes 37 105
Hydroalkylation of alkynes 37—38
Hydroboration see “Alkenes” “Alkynes”
Hydrobromic acid (aq. HBr), OH/Br exchange (aq.  ) 303 ) 303
Hydrobromic acid (aq. HBr), porphyrin synthesis with 255 349
Hydrobromination of alkynes with Zr 132 325
Hydrochlorothiazide 310
Hydrofluoric acid (aq. HF), deblocking with, of diol acetonides 277
Hydrofluoric acid (aq. HF), deblocking with, of silyl ethers 277 329
Hydrofluoric acid (aq. HF), deblocking with, of silyl thioethers 169
Hydroformylation of alkenes 46—48
Hydrogen peroxide, epoxidation with, of 2-enones (with KOH) 274—275
Hydrogen peroxide, epoxidation with, of alkenes (with HCOOH) 123
Hydrogen peroxide, oxidation with, of phenylselenyl groups 139
Hydrogen peroxide, oxidation with, of phosphines to P-oxides 282
Hydrogen peroxide, oxidative cleavage of 2-enones 82
Hydrogen peroxide, oxidative cleavage of phosphorus ylides 31
Hydrogen peroxide, oxidative cleavage or triorganylboranes 47—48 130—131 325
Hydrogen peroxide, oxidative rearr. (Baeyer — Villiger) of carbonyl compounds with  136—137 136—137
Hydrogen shift, sigmatropic 262 289 295 “Rearrangements sigmatropic”)
Hydrogenation 96—105 (see also “Hydrogenolysis”)
Hydrogenation of alkenes 41 96—97 101—103 278 283
Hydrogenation of alkynes to (Z)-alkenes 40 64 100—101
Hydrogenation of arenes 87 347
Hydrogenation of heteroarenes 112—113 303
Hydrogenation of iminium ions 293
Hydrogenation of nitroalkanes 66 112 297
Hydrogenation, catalysts, heterogeneous 96 100—102 105 111—113
Hydrogenation, catalysts, homogeneous 96—97 102—103 326
Hydrogenation, catalysts, homogeneous, chiral (Noyori) 102—103 325—326
Hydrogenation, mechanism 101—102
Hydrogenolysis 113—114 (see also “Reductive cleavage”)
Hydrogenolysis of activated keto groups 109
Hydrogenolysis of allylic alcohol derivs. 113—114 312—313
Hydrogenolysis of benzyl carbamates 143 162—163 229 240
Hydrogenolysis of benzyl ethers 157—159 161 271 328
Hydrogenolysis of benzylidene acetals 268
Hydrogenolysis of cyclopropane rings 105
Hydrogenolysis of haloalkanes 97 113—114 286—287
Hydrogenolysis of oxirane rings 113—114 319—320
Hydrogenolysis of thioethers 26 113—114
Hydrogenolysis of “Ox”-protected amines 161 164
hydrolysis see “Deblocking”
Hydrolysis, enantiotoposelective 167—168 276—277
Hydroperoxide, 1,1-dimethylethyl ( , tert-butyl hydroperoxide, TBHP), Sharpless epoxidation with 124—127 265 321—325 , tert-butyl hydroperoxide, TBHP), Sharpless epoxidation with 124—127 265 321—325
Hydrostannylation of alkynes 321
Hydroxamic acid chlorides = N-hydroxy carboximidoyl chlorides 153
Hydroxides, alkali, deprotonation with, of alcohols 157 159 265
Hydroxides, alkali, deprotonation with, of dicarbonyl type compounds 10 71—73
Hydroxides, alkali, deprotonation with, of hydrazones 109
Hydroxides, alkali, deprotonation with, of ketones 69 78 79 82 280
Hydroxides, alkali, deprotonation with, of oxosulfonium salts 8
Hydroxides, alkali, deprotonation with, of sulfonium salts 79 336
Hydroxides, benzyltrimethylammonium see “Benzene-methanaminium ...”
Hydroxy acids, chiral, pr. 176 183 185 191
Hydroxylamine-HCl, cleavage of acetonides 323
Hydrozirconation of alkynes 132 325
Hyperconjugation effects of alkyl groups on Baeyer — Villiger rearr. 136—137
Hyperconjugation effects of alkyl groups on enolate formation 11—13 57—58 93
Hyperconjugation effects of alkyl groups on relative reactivities of aldehydes vs. ketones to d-synthons 56
Hyperconjugation effects of alkyl groups on relative reactivities of alkenes to oxidation 87—88 123—124 156—157
Hyperconjugation effects of alkyl groups on relative reactivities of prim. vs. sec. alcohols to oxidation 135
Hypochlorites,  -catd. oxidation of alkynes 132 -catd. oxidation of alkynes 132
Hypochlorites, chlorohydrination with 117
Hypochlorus acid, 1,1-dimethylethyl ester, oxidative cleavage of thioethers with 314
Hypochlorus acid, 1,1-dimethylethyl ester, oxidative rearr. of 1-amino alcohols 77—78
Hypoiodites, acyl-, oxn. of alkenes with 127
Imidazoles, synthesis 151—152 305
Imides see “Dicarboximides”
Imidic acids see “Carboximidic acids”
Imido esters see “Carboximidic acids esters”
Imidogens see “Nitrenes”
Imines, N-alkyl- = N-alkylidene amines, hydrolysis of 57
Imines, N-alkyl- = N-alkylidene amines, N-acylation of 153
Imines, N-alkyl- = N-alkylidene amines, reduction of 112—113
Imines, N-alkyl- = N-alkylidene amines, synthesis of, by condensation of reactive 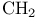 with nitroso compounds 308 with nitroso compounds 308
Imines, N-alkyl- = N-alkylidene amines, synthesis of, by trapping of diallylnickel complexes with isocyanides 41
Imines, N-alkyl-, lithiated 12 18 56—57 199
Imines, N-alkyl-, lithiated, directed aldol condensation with 56—57
Imines, synthesis of, aldehyde + amine 298
Imines, synthesis of, alkylation or reduction of nitriles 298—299
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of 1-alkynes 57
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of carboxamides 317—318
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of indoles and phenols 291—292
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of ketones 57 300 304
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of pyrroles 252
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), aminomethylation with, of sulfonamides 310
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), conversion to enamines 13 88 139
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), preparation, from carbonyl compounds 13 57 292 300 304 310 318
Iminium (= N-alkyl-N-alkylidene alkanaminium = immonium) ions (Mannich reagents), preparation, oxidation of tert. amines 116 120—121 138—139
Immunosuppressive agent “FK 506” 324—329
Indoles, 2-substitution, Friedel — Crafts type 291
Indoles, 2-substitution, Mannich type 292
Indoles, 2-substitution, Vilsmeier type 293
Indoles, N-acylation 307
Indoles, synthesis (Fischer) 151—152 296 307
Indomethacin 307
INOC = [(4-pyridinylmethoxy)carbonyl)] 241
Inserted DNA sequences 243—245
Insulin B-chain, chemical synthesis 239—240
Insulin, synth. by genetic engineering 244—245
Introns 242
Inversion of configuration see “Epimerization”
Inversion of configuration of alcohols (Mitsunobu reaction) 160—161 286
Inversion of configuration of alkyl halides or sulfonates by 1,3-dithiane anions 22
Inversion of configuration of alkyl halides or sulfonates by organylcuprates 36
Inversion of configuration of alkyl halides or sulfonates by tetracarbonyIferrate(2-) 46—47
Inversion of configuration of alkyl halides or sulfonates, in triorganylborane rearr. 37—38
Inversion of configuration of allylic acetates with Pd 27 164
Inversion of configuration of allylpalladium with d-synthons 27 264
Inversion of configuration of glycosides with free 2-OH by DAST 272
Inversion of configuration of glycosyl halides 271
Inversion of configuration of oxiranes by 1-alkyne anions 64 204
Inversion of configuration of oxiranes by enolate type anions 63—64
Inversion of configuration of oxiranes by internal alcoholate addition 265
Inversion-esterification of alcohols 160—161 286
Iodide, hydrogen, porphyrin synthesis with 255
Iodine = diiodine (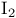 ) + ) + 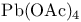 , ox. decomp. of RCOOH 337 , ox. decomp. of RCOOH 337
Iodine = diiodine (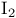 ) + carboxylic acids, oxidation of alkenes to 1,2-diols 127—128 ) + carboxylic acids, oxidation of alkenes to 1,2-diols 127—128
Iodine = diiodine (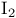 ) + hydroxide, rearr. of 1-alkenyl boranes 37 ) + hydroxide, rearr. of 1-alkenyl boranes 37
Iodine = diiodine (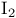 ) + triphenylphosphine, OH/I exchange 328 ) + triphenylphosphine, OH/I exchange 328
Iodine = diiodine (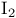 ) + water, oxn. of phosphites 219—220 223 ) + water, oxn. of phosphites 219—220 223
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



 ),
), 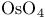 -catalysed oxn. with
-catalysed oxn. with 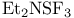 (DAST), OH/F exchange
(DAST), OH/F exchange 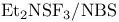 )
)  , oxidation of alkanes
, oxidation of alkanes 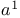 -synthon
-synthon 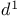 -synthons
-synthons 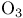
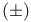 -
-  synthon
synthon  -elimination, 2-acetoxyglycals
-elimination, 2-acetoxyglycals  -, +
-, + 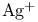 , activation of glycosyl fluorides
, activation of glycosyl fluorides 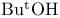 (Birch redn.)
(Birch redn.) 
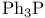

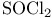 )
)  )
) 
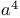 + alkenyl d
+ alkenyl d 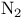 extrusion
extrusion  )
) 
 , tert-butyl hydroperoxide, TBHP), Sharpless epoxidation with
, tert-butyl hydroperoxide, TBHP), Sharpless epoxidation with  -catd. oxidation of alkynes
-catd. oxidation of alkynes  with nitroso compounds
with nitroso compounds 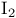 ) +
) + 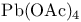 , ox. decomp. of RCOOH
, ox. decomp. of RCOOH