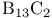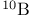Авторизация
Поиск по указателям
Greenwood N.N., Earnshaw A. — Chemistry of the Elements
Обсудите книгу на научном форуме Нашли опечатку?
Название: Chemistry of the ElementsАвторы: Greenwood N.N., Earnshaw A. Аннотация: When this innovative textbook first appeared in 1984 it rapidly became a great success throughout the world and has already been translated into several European and Asian languages. Now the authors have completely revised and updated the text, including more than 2000 new literature references to work published since the first edition. No page has been left unaltered but the novel features which proved so attractive have been retained. The book presents a balanced, coherent and comprehensive account of the chemistry of the elements for both undergraduate and postgraduate students. This crucial central area of chemistry is full of ingenious experiments, intriguing compounds and exciting new discoveries. The authors specifically avoid the term `inorganic chemistry' since this evokes an outmoded view of chemistry which is no longer appropriate in the final decade of the 20th century.
Язык: Рубрика: Химия /Статус предметного указателя: Готов указатель с номерами страниц ed2k: ed2k stats Издание: second editionГод издания: 1997Количество страниц: 1340Добавлена в каталог: 18.02.2007Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
Предметный указатель
Argon, production and uses 889 Arsenates 577 Arsenic, abundance and distribution 548 549 Arsenic, allotropes 551 Arsenic, alloys 549 554 Arsenic, amino derivatives 561 Arsenic, atomic properties 550 Arsenic, catenation 583—590 Arsenic, chalcogenide cluster cations 579 Arsenic, chalcogenides 578—583 Arsenic, chemical reactivity 552—554 577 Arsenic, cluster anions 553 588—591 Arsenic, coordination geometries 553 Arsenic, diiodide 564 Arsenic, encapsulated 554 Arsenic, extraction and production 548 549 Arsenic, halide complexes 564—569 Arsenic, halides 558—566 see trihalides” “Arsenic pentahalides” “Arsenic diiodide” “Arsenic oxide “Arsenic mixed “Arsenic halide Arsenic, halogenoorganoarsenic compounds 593—596 Arsenic, history 547 Arsenic, hydrides 557 558 583 679 Arsenic, intermetaUic compounds see “Arsenides Arsenic, metal-metal bonded species 583—590 Arsenic, mixed halides 563 Arsenic, organic compounds 553 Arsenic, organoarsenic(I) compounds 597 Arsenic, organoarsenic(III) compounds 583—587 592—594 596 Arsenic, organoarsenic(III) compounds, arsabenzene 593 Arsenic, organoarsenic(III) compounds, arsanaphthalene 593 Arsenic, organoarsenic(III) compounds, physiological activity 596 Arsenic, organoarsenic(III) compounds, preparation of 593 595 Arsenic, organoarsenic(III) compounds, reactions of 593 595 Arsenic, organoarsenic(V) compounds 594 596 Arsenic, oxidation state diagram 578 Arsenic, oxide 549 572—575 Arsenic, oxide 574 Arsenic, oxide 573 Arsenic, oxide 549 574 Arsenic, oxide halides 570—572 Arsenic, oxides and oxocompounds 570—578 Arsenic, oxoacid salts of 591 Arsenic, pentahatides 561. 664 Arsenic, physical properties 551 552 Arsenic, selenides 581 Arsenic, sulfide, 547 548 578 579 648 Arsenic, sulfide, 580 581 Arsenic, sulfide, 578 579 Arsenic, sulfide, 548 578—581 649 Arsenic, sulfides, 578—580 Arsenic, triangular species 583 586—589 Arsenic, trichloride solvent system 560 Arsenic, trihalides 558—561 564 566 Arsenic, uses 549 Arsenic, volt-equivalent diagram 578 Arsenicals, therapeutic uses 593 see Arsenides 554—558 Arsenides, stoichiometries 554 Arsenides, structures 554—557 Arsenidine complexes 597 Arsenious acid 574 Arsenite 575 575 577 Arsenite esters 561 Arsenopyrite 649 Arsine 558 Arsinic acids 594 Arsinous acids 594 Arsonic acids RAsO(OH) 596 Arsonous acids 594 Asbestos 109 351 Asbestos minerals 349 351 Astatate ion, 886 Astatide ion 886 Astatine see “Halogens” Astatine, abundance 796 Astatine, atomic properties 800 Astatine, chemistry 885. 886 Astatine, nuclear synthesis 791 795 Astatine, radioactivity of isotopes 795 885 Astatine, redox systematks 885 Astatine, trihalide ions 886 Atactic polymer 972 Atmophile elements 648 Atmosphere, composition 409 603 Atmosphere, industrial production of gases from 409 604 Atmosphere, origin of 602 Atom-at-a-time chemistry 1282 Atomic energy 1256 Atomic orbitals 1285—1289 Atomic piles 1256 see Atomic properties, periodic trends in 23—27 Atomic volume curve, periodicity of 23 24 Atomic weight, definition 15 Atomic weights, history of 15 601 Atomic weights, precision of 15—19 Atomic weights, Table of see “Front end paper” Atomic weights, variability of 17—19 368 ATP see “Adenosine triphosphate” Autoprotolysis constants of anhydrous acids 710 Azides 433 Azoferredoxin 1036 1098 Azotobacter 999 1036 Azotobacter vinetandii 1036 Back ( 923 926 927 931 1166 Baddeleyite 955 Baddeleyite, structure of 955 Baking powders 524 525 Barium see “Alkaline earth metals” Barium, history of 108 Barium, organometallic compounds 136 Barium, polysulfides 681 Bart reaction 596 Basic oxygen steel process 120 1072 Bastnaesite 945 1229 1232 Batteries, dry 1204 Batteries, lead 371 549 Batteries, sodium-sulfur 678 Bauxite 243 Bauxite in Al production 219 Bauxite, production statistics 218 Bayer process 167 Bayerite 243 245 Belousov — Zhabotinskii oscillating reactions 865 Bentonite see “Montmorillonite” Benzene as ligand 940—941 Berkelium 1252 1262 see Berry pseudorotation 474 499 914 see Beryl 107 108 349 Beryllia see “Beryllium oxide” Beryllium see “Alkaline earth metals” Beryllium compounds, toxicity of 107 Beryllium ion, hydration number of 605 Beryllium, alkoxides etc 122 129 Beryllium, alkyls 126—130 Beryllium, borohydridc 115 116 Beryllium, chloride 116 117 Beryllium, complexes of 122—125 Beryllium, cyclopentadienyl complexes 130 Beryllium, discovery 107 108 Beryllium, fluoride 116 Beryllium, hydride 115 115 128 Beryllium, oxide 107 119—120 Beryllium, salts, hydrolysis of 121 Beryllium, uses of metal and alloys 110 Beryllium, ‘anomalous’ properties of 114 122 Beryllium, ‘basic acetate’ 122 123 Beryllium, ‘basic nitrate’ 122 Bessemer process 1072 BHB bridge bond, comparison with H bond 64 70 see Biotite see “Micas“ Bismuth, abundance and distribution 548 549 550 Bismuth, allotropes 551 551 Bismuth, alloys 549 554 Bismuth, atomic properties 550 Bismuth, Bi+cation 564 591 Bismuth, catenation 582 Bismuth, chalcogenides 581 582 Bismuth, chemical reactivity 552 553. Bismuth, cluster cations 564 565 583 588—591 Bismuth, extraction and production 549 550 Bismuth, halide complexes 564—567 Bismuth, halides 558—564 see trihalides “Bismuth pentahalides” “Bismuth oxide “Bismuth lower “Bismuth mixed “Bismuth halide Bismuth, history 547 Bismuth, hydride 557 558 Bismuth, hydroxide, 575 Bismuth, hydroxo cluster cation, 575 591 592 Bismuth, intermetallic compounds see “Bismuthides” Bismuth, lower halides 564 Bismuth, metal-metal bonded clusters 583 583—591 Bismuth, mixed halides 564 Bismuth, nitrate and related complexes 591. 592 Bismuth, organometallic compounds 592 596 599 Bismuth, oxidation state diagram 578 Bismuth, oxide halides 572 572 Bismuth, oxide, 573 574 Bismuth, oxides and oxo compounds 573—575 Bismuth, oxoacid salts of 591 Bismuth, pentafluoride 561 Bismuth, physical properties 550 552 Bismuth, trihalides 558—561 564 566 Bismuth, uses 549 Bismuth, volt-equivalent diagram 578 Bismuthates 577 Bismuthides 554 Bismuthine 557 Biuret 305 1191 Blast furnace 1073 Bleaching powder 790 860 Blue vitriol 1190 Boehmite 243 Bohrium 1281 1283 Boiiing points, influence of H bonding 54 Bom — Haber cycle 94—96 79 82—83 Borane adducts, 165 Borane adducts, amine-boranes 208—209 Borane anions 151 166 178 181 590 see Boranes, arachno-structures 152 154 159 Boranes, as ligands 177 Boranes, bonding 157—162 590 Boranes, classification 151 Boranes, closo-structures 152 153 161 Boranes, conjuncto-structures 152 155—157 Boranes, hypno-strucuires 152 153 172 Boranes, nido- structures 152 154 161 Boranes, nomenclature 157 Boranes, optical resolution of 670 Boranes, physical properties 162 163 Boranes, preparation 162 Boranes, topology 158—160 175 see “Petaborane” “Decaborane” “Metalloboranes” “Carboranes” etc. Borate minerals, occurrence 140 Borate minerals, production and uses 140 see Borates 205—207 Borates, B-O distances in 206 Borates, industrial uses 207 Borates, structural principles 205 Borax 139 see Borazanes 209 Borazine 210 408 Bordeaux mixture (fungicide) 1190 Boric acids, 203 Boric acids, 204 Borides, bonding 151 Borides, catenation in 148 Borides, preparation 146 Borides, properties and uses 146 Borides, stoichiometry 145 147 Borides, structure 147—151 Borohydrides see “Borane anions” “Tetrahydro-borates” Boron 167 see Boron carbide 149 Boron carbide 149 Boron carbide 143 Boron halides 195—202 Boron halides, 200 202 Boron halides, 200 Boron halides, lower halides 199—202 see Boron nitride, $\mathrm{B_{50}N_2} 143 Boron trifluoride see “Boron trihalides” Boron trihalides, adducts 198—199 Boron trihalides, bonding 196 Boron trihalides, physical properties 196 Boron trihalides, preparation 196 Boron trihalides, scrambling reactions 197 198 Boron, abundance 139 Boron, allotropes 141—144 Boron, atomic properties 144 222 Boron, chemical properties 144 145 Boron, crystal structures of allotropes 141 Boron, hydrides see “Boranes” Boron, isolation 139—140 144 Boron, neutron capture therapy using 179 Boron, nitride 208 208 Boron, nuclear properties 144 Boron, oxide 203 Boron, physical properties 144 222 Boron, sulfides 213—214 Boron, variables atomic weight of 17 Boron-nitrogen compounds 207—211 Boron-oxygen compounds 203—207 Boron-oxygen compounds, organic derivatives 207 brass 1175 1178 1178 1201 1203 1397 Brimstone 645 646 Bromates, 862—864 Bromates, 866 Bromates, 853—856 Bromic acid, 863 Bromides, synthesis of 821 822 see Bromine see “Halogens” Bromine, abundance and distribution 795 Bromine, atomic and physical properties 800—804 Bromine, cations 842—844 Bromine, history 790 790 792. 925 Bromine, monochloride 824 825 833 Bromine, monofboride 825 833 Bromine, oxide fluorides 880—881 Bromine, oxides 850 851 Bromine, oxides, nomenclature 853 Bromine, oxides, redox properties 853—855 see oxides individual “Bromine oxides bromic “Bromine oxides bromates” “Bromine oxides perbromates” etc. Bromine, pentafluoride 832—834 Bromine, production and uses 798 800 Bromine, radioactive isotopes 801 802 Bromine, reactivity 805 Bromine, standard reduction potentials 854 Bromine, stereochemistry 806 Bromine, trifluoride 827—831 832 Bromine, volt-equivalent diagram 855 Bronsted's acid-base theory 32 48 Bronsted's acid-base theory in aqueous solutions 628 Bronsted's acid-base theory in non-aqueous solutions 51 Bronze 1173 1175 Bronzes, molybdenum 1016 Bronzes, titanium 964 Bronzes, tungsten 1016 Bronzes, vanadium 987 Brucite 121 352 385 Buffer solutions 48 49 521 524 Bulky tertiary phosphine ligands, special properties of 494 Butadiene as a fj4 ligand 935—936 Cadmium chloride structure 1211 Cadmium iodide structure type 556 680 680 1211 Cadmium iodide structure type, nonstoinchiometry in 679
Реклама
 |
|
О проекте
|
|
О проекте



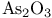
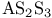
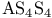



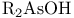
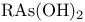
 "
" 
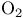 in
in  ) bonding
) bonding 
![$\mathrm{[Bi_6(OH)_12]^{6+}}$](/math_tex/80e0ee6dc34d7960239e6a68f95c6b2382.gif)