|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Galwey A.K. — Thermal Decomposition of Ionic Solids |
|
|
 |
| Предметный указатель |
Abnormal reactions 123—124
Absolute reaction rate theory 117 124—125 133—134
Absolute reaction rate theory, hydroxyhalide decompositions 375
Absolute reaction rate theory, potassium permanganate decomposition 382
Acceleratory kinetic rate equations 102—111
Acceleratory period 106—110 142
Acetate ion, decomposition 450—451
Acetates, metal, decompositions 448—451
Acid metal fluorides, decompositions 374—375
Activated complex 117 124—126
Activation energies for metal oxalate decompositions 468
Activation energies, oxyhalide decompositions 376
Activation energy 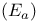 117—134 199 567G 117—134 199 567G
Activation energy, band structure, chromate decomposition 389—390 404
Activation energy, carbonate decomposition 345—346
Activation energy, nickel carboxylate decomposition 483
Activation energy, perchlorate decomposition 366
Active reaction zone 126—129
Additives and silver oxalate decomposition 457
Additives in lead azide decomposition 336—337
Additives, specific 192
Adsorbed species 47—48
Adsorbed species in metal promoted reactions 544
Adsorbed species, mobile 125
Adsorbed water 229 241
Alkali metals see “Group IA metals”
Alkaline earth metals see “Group IIA metals”
Alpha ( ) see also “fractional reaction” 30 60 570G ) see also “fractional reaction” 30 60 570G
Alum dehydrations 236—241 251
Aluminium hydride, decomposition 316 533
Aluminium hydride, irradiation 316
Aluminium hydroxides, dehydration 273—275
Aluminium sulfate hydrate, dehydration 235
Aluminium sulfate, decomposition 401—402 405
Ammine chromium(III) halides and nitrate, decompositions 504—506
Ammine cobalt(II) halides, nitrate and sulfate, decompositions 503
Ammine cobalt(III) azides,decompositions 500—502 521
Ammine cobalt(III) hydrate salts, decompositions 500
Ammine cobalt(III) iodides, decompositions 498—499
Ammine nickel coordination compounds, decompositions 507—508
Ammine palladium coordination compounds, decompositions 507
Ammine platinum coordination compounds, decompositions 507
Ammonia decomposition, iron nitride 320
Ammonium aluminium sulfate, basic, decomposition 426
Ammonium azide, thermal reactions 431
Ammonium bicarbonate, decomposition 431
Ammonium bromate, decomposition 423
Ammonium carbonate, decomposition 431
Ammonium carboxylates, decompositions 431—432
Ammonium chlorate, decomposition 423
Ammonium chromate, decomposition 428—430
Ammonium copper sulfate hexahydrate, dehydration 229
Ammonium dichromate, decomposition 429—430
Ammonium dichromate, decomposition, melting 430
Ammonium halates, decompositions 422—423
Ammonium iodate, decomposition 423
Ammonium molybdates, decompositions 432
Ammonium nitrate in ammonium salt decompositions 423 429—430 434—435
Ammonium nitrate, decomposition 424
Ammonium oxalate monohydrate, dehydration 248
Ammonium perchlorate, decomposition, catalyzed 419—420
Ammonium perchlorate, decomposition, high temperature 415—420
Ammonium perchlorate, decomposition, low temperature 415—420 508
Ammonium perchlorate, decomposition, mechanisms 418—420
Ammonium perchlorate, dislocations 416—417
Ammonium perchlorate, microscopy 416—417
Ammonium perchlorate, nucleation 416
Ammonium perchlorate, pre-irradiation 417
Ammonium perchlorate, proton transfer 417—419
Ammonium perchlorate, unified reaction scheme 417—418
Ammonium permanganate, decomposition 430—431
Ammonium permanganate, irradiation 431
Ammonium peroxovanadate, decomposition 428
Ammonium perrhenates, decompositions 432—433
Ammonium phosphate, anion condensation 425
Ammonium phosphate, decompositions 425—426
Ammonium phthalates, decompositions 432
Ammonium salt decompositions, sublimation 418—419 431 434
Ammonium salts with oxidizing anions, decompositions 545
Ammonium salts, decompositions 415—435
Ammonium salts, decompositions, nitrate formation 545
Ammonium salts, decompositions, proton transfer 419 423—425 428 434
Ammonium sulfate, decomposition 399 426
Ammonium tungstates, decompositions 432
Ammonium uranates, decompositions 433—434
Ammonium vanadates, decompositions 427—428
Anation reactions in coordination compounds 500 506 510—511 518 522
Anionic electron transfer, potassium permanganate 383
annealing 24 32 51 571G
Annealing and imperfection loss 558
Annealing, silver oxide 299
Aragonite-calcite transition 345—346
Arrhenius equation 48 67 117—134 567G
Arrhenius parameters 118—125 148—149 153—156 163—164 222 247 282 284
Arrhenius parameters, anomalous 223
Arrhenius parameters, decompositions of solids 529 541—542 551 557 559
Arrhenius parameters, normal 223 237
Arrhenius parameters, reactant area 190
Arrhenius plots 122—123 144—145 163
Atmosphere, influence on kinetic measurements 535—537
Auger electron spectroscopy 177—178
Austin — Rickett kinetic rate equation 95—96
Autocatalysis in solid decompositions 557
Autocatalytic reactions 94—96
Avrami — Erofeev equation exponents 144
Avrami — Erofeev rate equation see also “JMAEK equation” 89 91 96 99 103 107—108 542
Azide decomposition, exciton formation 341
Azide decomposition, radiation generation 340
Azides, band structures 329—330
Azides, chemical and physical properties 330
Azides, cobalt(III) ammines, decompositions 500—502 521
Azides, conductivites 329
Azides, crystal imperfections 329—330
Azides, crystal structures 330
Azides, decompositions 329—341
Azides, dissociative evaporation 340
Azides, Group IIA metal, decompositions 331—333
Azides, nucleation and growth 329
Azides, optical properties 329
Azides, radiolysis 329—330
Band structure, chromate decompositions 389—390 404
Band structure, interface 127—129
band theory 6 17—22
Barium acetate, decomposition 450
Barium azide, decomposition 331—333 339—340
Barium bromate, decomposition 372
Barium carbonate, decomposition 350—351 358
Barium chloride hydrates, dehydration 242
Barium chlorite, decomposition 374
Barium copper oxalate, decomposition 466
Barium hydrogen oxalate hydrate, dehydration 248
Barium iodate, decomposition 372
Barium nitrite, decomposition 395
Barium oxalate dihydrate, dehydration 248
Barium oxalate, decomposition 462—463
Barium perchlorate, decomposition 367
Barium permanganate, decomposition 388
Barium permanganate, decomposition, irradiation 388
Barium peroxide, decomposition 298—299
Barium styphnate trihydrate, dehydration 252
Barium styphnate, decomposition 477
Barium sulfate, decomposition 399 405
Barium titanyl oxalate, decomposition 466
Barrier layer, product 96—100
Beryllium hydride, decomposition 315
Beryllium hydroxide, dehydration 273
Beryllium sulfate, decomposition 400 405
Bidentate ligands 512
Bimolecular reactions 125
Binary compounds, decompositions 313—325
| Binary compounds, stabilities 323
Binary compounds, sublimation 323—325
Binuclear coordination compounds, decompositions 517
Binuclear coordination compounds, formation 511
Block structures 306—307
Bond activation 129
Bond redistribution steps (Table 18.1) 126—127 133—134 199 200 530—532 542
Borate hydrate, dehydration 244—245
Borax, dehydration 244
Borchart and Daniels method of kinetic analysis 159—160
Bose — Einstein statistics 127—128
Boundary layer, surface 76
Bubble development 230—231
Burger's vector 14 24
Cadmium acetate, decomposition 450
Cadmium basic halides, decompositions 376
Cadmium carbonate, decomposition 355 358
Cadmium cyanamide, decomposition 338
Cadmium hydroxide, dehydration 276
Cadmium hydroxide, sintering and impurities 276
Cadmium hydroxide, unreacted crystals 276
Cadmium iodide, decomposition 374
Cadmium oxalate, decomposition 463—464
Cadmium oxide, decomposition 301
Cadmium sulfide, sublimation 44
Caesium azide, decomposition 335
Caesium bromate, decomposition 371 374
Caesium graphite, decomposition 319—320
Caesium orthophosphate, decomposition 397
Caesium periodate, decomposition 370
Caesium permanganate, decomposition 383 386—387
Cage effect 47
Calcite see “calcium carbonate”
Calcium acetate, decomposition 450
Calcium azide, decomposition 333 339
Calcium carbonate, decomposition 132—133 346—349 357—358 360 533 547
Calcium carbonate, decomposition, Arrhenius parameters 347—348
Calcium carbonate, decomposition, diffusion 348
Calcium carbonate, decomposition, heat transfer 348
Calcium carbonate, decomposition, reversibility 346—349
Calcium carbonate, finely divided calcium oxide 346 348
Calcium carbonate, impurities 346—347
Calcium carbonate, nucleation and dislocations 346—347 360—361
Calcium carbonate, reaction interface 346 360
Calcium fumarate, decomposition 475 485
Calcium hydrogen phosphate, decomposition 397
Calcium hydroxide, dehydration 272—274
Calcium iodate, decomposition 372
Calcium maleate, decomposition 475 485
Calcium malonate, decomposition 475 485 542—543
Calcium nitrate, decomposition 392—394
Calcium nitrate, decomposition, metal oxide catalyzed 393—394
Calcium oxalate hydrate, dehydration 247—248 559
Calcium oxalate, decomposition 462—463 486
Calcium oxide from calcite decomposition 547
Calcium perchlorate, decomposition 368
Calcium peroxide, decomposition 298
Calcium propionate, decomposition 451
Calcium sulfate dihydrate, dehydration 232—234
Calcium sulfite, decomposition 402—403
Calcium-magnesium oxide from dolomite decomposition 350
Carbide decompositions 317—320
Carbonates, decompositions 345—361
Carbonates, Group IA metal, decomposition 351—352
Carbonates, Group IIA metal,decompositions 345—351 358—361
Carboxylate decomposition, reactivity patterns 482—486
Carboxylate decomposition, residual solid 481—482
Carboxylate decompositions, controlling bond rupture step 479—480
Carboxylate decompositions, dehydration 478
Carboxylate decompositions, electron transfer 443 457 462 480
Carboxylate decompositions, hydrolysis 478
Carboxylate decompositions, mechanisms 479—480 544 557
Carboxylate decompositions, secondary reactions 479
Carboxylates, ammonium, decompositions 431—432
Carboxylates, metal, decompositions 54—55 441—486
Carboxylates, nickel, decompositions 475—476
Carroll and Manche method of kinetic analysis 157
Catalysis, metal product 544 557
Catalyst preparation 282
Catalysts, mixed oxides, permanganate decomposition 389
Catalytic decomposition, sodium and potassium chlorates 370
Chemical features, decompositions of solids 528 554
Chemical foundations of solid state reactions 202
Chemisorbed species 126
Chemisorption, oxygen on oxides 291
Chlorates, Group IA metal, decompositions 370 374
Chlorates, Group IIA metal, decompositions 371
Chlorates, metal, decompositions 370—371 374
Chlorites, metal, decompositions 373
Chromates, decompositions 389—390 404
Chrome alum, dehydration 236 253
Chromium hydride, decomposition 314—315
Chromium hydroxide, dehydration 277—278
Chromium hydroxide, topotactic oxidation 277—278
Chromium oxides, decompositions 302—303
Chromium sulfate, decomposition 400
Chromium(III) ammines, decompositions 504—506
Chromium(III) ethylenediamine compounds, decompositions 513—514
Chromium(III) oxalato ammine compounds, decompositions 516
Chromium(III) tris-N-benzoyl-N-phenylhydroxylamine, decomposition 506
Citrate, lead, decomposition 476—477
Classification, dehydrations of solids, (Figure 7.4) 258—259
Classification, reactions of solids 527—528 533 555 558—560
Clay dehydrations 283—287
Clays, catalytic reactions 284
Cleavage 15
Cleaved crystal faces 237
Cobalt acetate, decomposition 449
Cobalt acetate, hydrolysis 449
Cobalt carbide, decomposition 318—319
Cobalt carbonate, decomposition 356
Cobalt fluoride, decomposition 374
Cobalt hydroxide, dehydration 280
Cobalt iodate, decomposition 373
Cobalt malonate, decomposition 469 542—543
Cobalt oxalate, decomposition 454—455 485—486
Cobalt oxide, decomposition 304
Cobalt oxide, reduction 304—305
Cobalt phosphate hydrate, dehydration 397
Cobalt sulfate binuclear compound, decomposition 517
Cobalt sulfate, decomposition 401 406
Cobalt sulfide, decomposition 322
Cobalt(II) ammines, decompositions 503
Cobalt(II) pyridine halides, decompositions 503—504
Cobalt(III) ammine azides, decompositions 545 555
Cobalt(III) ammines, decompositions 495—502 515
Cobalt(III) bis(dioximato) compounds, decompositions 517
Cobalt(III) coordination compounds, perchlorate, decompositions 513
Cobalt(III) diammine compounds, decompositions 512—513
Cobalt(III) diammine compounds, dehydration 513
Cobalt(III) ethylenediamine compounds, decompositions 512—513
Cobalt(III) hexaammine halides, nitrate and sulfate, decompositions 495—499
Cobalt(III) oxalato ammine compounds, decompositions 515
Cobalt-nickel mellitate, decomposition 467
Coherent intergrowth, oxides 307
Colour centre 11
Compensation effect 48 129—134 235 273 284—285 293 541—542 554—559 569G
Compensation effect and reversibility 542
Compensation effect in coordination compound decompositions 517
Complex compounds 493
Concentrations 1 568G
Concurrent reactions 163—164
Concurrent reactions in decomposition 547
Conditions of reactions and coordination compounds 519 521
Conductivity and silver oxalate decomposition 456
Conductor 16—22
Consecutive reactions 164
Constant rate thermal analysis (CRTA) 148
Contracting area or disc rate equation 93 104
Contracting cube or volume rate equation 92 98—99 101—104 108—110
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



 )
)