|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Galwey A.K. — Thermal Decomposition of Ionic Solids |
|
|
 |
| Предметный указатель |
Controlled atmosphere 221
Controlling step, decompositions of solids 528 554 557—560
conversion functions 567G
Coordinated water 217
Coordination bond rupture 530 532
Coordination compound 493
Coordination compounds, decomposition mechanisms 494—495
Coordination compounds, decompositions 493—522
Coordination compounds, hydrates as 494
Coordination compounds, thermal stabilities 517—520
Coordination number 493
Coordination polyhedron 494
Copper acetate, decomposition 449
Copper ammine chromate, decomposition 509
Copper ammine sulfate, decomposition 509
Copper azide, decomposition 335 339
Copper carbonate, decomposition 356
Copper carboxylate, decomposition, metal sublimation 444 449
Copper carboxylate, decompositions, copperВ® intermediates 43 449 458 469 473 484
Copper chloride piperazinium hydrate, dehydration 509
Copper chloride pyrimidinium hydrate, dehydration 509
Copper formate tetrahydrate, dehydration 249
Copper formate, decomposition 442—445 448 484
Copper formate, decomposition mechanism 443—445
Copper formate, decomposition, copper(1) intermediate 443—444
Copper fumarate, decomposition 473 484
Copper hexaammine halides, decompositions 508—509
Copper hydride, decomposition 316
Copper hydroxide, dehydration 281
Copper hypophosphite, decomposition 398
Copper maleate, decomposition 473 484
Copper malonate, decomposition 472—474 484 542—543
Copper nitrate, decomposition 394
Copper nitride, decomposition 320
Copper oxalate, decomposition 457—458 485—486
Copper oxides, decomposition 300
Copper permanganate, decomposition 389
Copper squarate, decomposition 469 484
Copper sublimation in copper carboxylate decompositions 444 449
Copper sulfate pentahydrate, dehydration 123 223 227—229
Copper sulfate trihydrate, dehydration 227—229
Copper sulfate, decomposition 401—402 407
Copper sulfide, decomposition 322
Copper(II) salts, cation reductions 545
Copper(II) salts, decompositions 545
Copper, basic halides, decompositions 376
Covalent bond rupture 530 544
Covalent crystal decompositions 531
Crack development 96 98 111 234
Cracking, potassium bromate decomposition 371
Cracking, potassium metaperiodate decomposition 369
Cracking, potassium permanganate reaction 381—382
Crystal bulk 569G
Crystal cracking 270 274 276 282
Crystal imperfections 8—15
Crystal imperfections in decompositions of solids 526 529 556
Crystal lattice 2
Crystal strain 271
Crystal structure 174—175
Crystal structures and stabilities 555
Crystal surface 569G
Crystal, charge-carrying species 191
Crystalline hydrates 217
Crystallographic shear 306—307
Crystals, covalent 5
Crystals, ionic 5
Crystals, metallic 6
Crystals, molecular 5
Crystolysis 3 111 527 537 569G
Crystolysis and related reactions 551—552
Crystolysis reactions, literature content 548
Cyano coordination compounds, decompositions 510—511
Deceleratory kinetic rate equations 102—111
Deceleratory period 106—110 143
Decomposition enthalpy 60 66
Decomposition of solids, steps 536 554
Decomposition products 53 55—56 58 62
Decomposition products, primary 53—54
Decomposition, classification of mechanisms 200—202 207—208
Decomposition, oxides 291—308
Decomposition, reversible, enthalpies 60
Decomposition, stoichiometry 53 60 63—64
Decompositions 25—26
Decompositions of coordination compounds 493—522
Decompositions of solids, chemical features 528 554
Decompositions of solids, controlling step 528 554 557
Decompositions of solids, reaction precursors 529 554
Decompositions with melting 547 554
Decompositions, ammonium salts 415—435
Decompositions, carbonates 345—361
Decompositions, dissociation pressure 52
Decompositions, endothermic 50 53 66—68
Decompositions, exothermic 50 66
Decompositions, Group IA metal permanganates 546 552
Decompositions, irreversible 200
Decompositions, irreversible, oxyacid salts 404
Decompositions, metal carboxylates 441—486
Decompositions, metal formates 441—448
Decompositions, metal salts of organic acids 441—486
Decompositions, oxyacid metal salts 381—409
Decompositions, oxyhalide salts 365—377
Decompositions, reversible 50 60 200
Decompositions, reversible, oxyacid salts 404
Defect mobility 16—25
Dehydration 31 217—268
Dehydration and cobalt oxalate decomposition 454
Dehydration kinetics 217—268
Dehydration mechanisms 218 256—260
Dehydration nuclei 221—222 258—259
Dehydration nuclei, shapes 232 237
Dehydration of 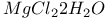 and oxidation of and oxidation of 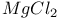 546 546
Dehydration, calcium oxalate monohydrate 559
Dehydration, experimental 219—221
Dehydration, explosive 261
Dehydration, Group IA metal cyanoferrates 510
Dehydration, hydroxides 269—290
Dehydration, initial 65
Dehydration, isotope effects 261
Dehydration, reversible 269
Dehydration, topotactic 257—260
Dehydration-anation reactions 260
Dehydration-rehydration 233
Dehydrations in mixed coordination compounds 500 506 517 522
Dehydrations,  and and  546 546
Dehydrations, classifications 256—259
Dehydrations, cobalt(III) diammine compounds 513
Dehydrations, copper chloride coordination compounds 509
Dehydrations, homogeneous 257
Dehydrations, hydroxyhalides 375—376
Dehydrations, lignite and alums 546
Dehydrations, lithium potassium tartrate hydrates 547
Dehydroxy lation reactions 281—2
Dehydroxy lation, hydroxides 269—290
Dehydroxy lation, inhomogeneous 270
Dehydroxylation, electric field 271
Dehydroxylation, homogeneous 270
Dense initial nucleation 234
Desorption, caesium from graphite compound 320
Desorption, oxygen 292
Diammine cobalt(III) compounds, decompositions 512—513
Diammine isomerization in solid coordination compounds 513
Differential scanning calorimetry (DSC) 63 66
Differential thermal analysis (DTA) 63
Diffraction measurements 537 554
Diffraction using synchrotron radiation 537 553
Diffusion 75 96—100 282 284
Diffusion coefficients 22 99
Diffusion control rate equations 96—111
Diffusion in potassium metaperiodate decomposition 369
Diffusion processes, hydroxyhalide decompositions 375
| Diffusion reactions 96—101
Diffusion, Fick's laws 97—99
Diffusion, intracrystalline 96—100 102
Diffusion, lattice 292
Diffusion, surface 43
Diffusion, water 231 242 250 258
Diffusion, water vapour 243 251
Dislocation detection, X-ray topography 184—185
Dislocation generation in sodium chlorate 370
Dislocations 9 12—15 24 37—38 42 46
Dislocations, Burger's vector 382
Dislocations, edge 12—13
Dislocations, screw 12—14
Dissociation, surface 42
Dissociations 568—569G
Dissociations, oxides 291—308
Dissociative evaporation, azides 340
Dissociative vaporization, equimolar 308
Dissociative vaporization, isobaric 308
Dissolution-recrystallization 279
Distinguishability of kinetic equations 141 143—144 147 149 540
Dolomite, decomposition 350 358
Dopants in barium bromate decomposition 372
Doping 12
Doping of crystals 192—193
Doping, cadmium carbonate decomposition 355
Doping, cobalt oxide 304
Doping, silver carbonate decomposition 354
Dunwald — Wagner rate equation 99
Elastic surface layer 230—231
Electrical conductivity 38 190—192
Electron diffraction 277
Electron mobility 16 22—25
Electron probe microanalysis 184
Electron spin resonance spectroscopy (ESR) 181
Electron transfer 44 127 530 532 544
Electron transfer in complex azide decompositions 502
Electron transfer, carboxylate decompositions 443 457 462 480
Electron transfer, oxides 291
Electron traps, 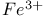 ions in barium azide 332 ions in barium azide 332
Electronic activation 75
Electronic energy 128 134
Electrons and positive holes 8—10 16—22
Ellingham diagrams 55—58
Energy band levels 191 199
Energy chain branching 381—382
Energy chains 79—80 96
Energy distributions, interface 127—129
Energy levels, spectral studies 534 538 559
Enthalpy of decomposition 60 69
Enthalpy of dehydration 222—223
Etch pits 14 42 230
Ethylenediamine chromium(III)compounds, decompositions 513—514
Ethylenediamine cobalt(III) compounds, decompositions 512—513
Ethylenediamine nickel compounds, decompositions 514
Evolved gas analysis (EGA) 61 539
Exciton formation in azide decomposition 341
excitons 9—10 20—21
Experimental conditions 140
Experimental errors 141 162 167
Experimental methods 29 60—67 69 174—194 277—278
Experimental methods, spectroscopic 177—178
Exponential rate equations 94 103
Extended imperfections 306—308
Extended X-ray absorption fine structure (EXAFS) 182—183
Extent of reaction (a) see also “fractional reaction” 30 60
F-centre 11
Fermi level 128
Fermi level, azide decompositions 332 334 340
Fermi — Dirac statistics 127—128
Fick's laws of diffusion 22
Field-emission spectroscopy 177—178
First-order rate equation 90—91 100 108
Flynn and Wall method of kinetic analysis 160
Flynn method of kinetic analysis 157
Formates as heterogeneous catalysis intermediates 447—448
Formates, metal, decompositions 441—448
Formates, metal, hydrates, dehydrations 249—250
Formic acid, nickel catalyzed decomposition 441—442 447
Fractional reaction (a) 30 60 87 139—140 570G
Frank net 14
Freeman and Carroll method of kinetic analysis 158
Frenkel point defects 9—11 24
Frenkel-type defects in dehydration 511
Frequency factor (A) 117—134 222 567G
Friedman method of kinetic analysis 157
Fulminates, decompositions 338—9
Fumarates, metal, decompositions 469 471—475
G thiosulfates, decompositions 403
Gas analyses 205
Gas chromatography (GC) 62
Gas pressure, accumulatory 61
Geometric and diffusion control rate equations 98—100
Geometric interpretation of kinetics 141 165
Geometric interpretations, microscopy 186—190
Geometric rate equations 91—94 102—111
Germ nucleus 77—78
Ginstling — Brounshtein rate equation 98
Glass 4
Glass transition 67
Glossary of subject-specific terms 567—571
Grain boundaries 8 24
Group IA and Group IIA metal oxides 297
Group IA metal formates, decompositions 446—447
Group IA metal hexacyanoferrates, decomposition 510
Group IA metal percarbonates 254—255
Group IA metal peroxocarbonates 254—255
Group IIA metal acetates, decompositions 450
Group IIA metal formates, decompositions 447
Group IIA metal hydrides, photodecomposition 315
Growth nucleus 25 76—78 239 258—259
Growth, restricted, nuclei 85—86
Growth, unrestricted, nuclei 84—85
Halides, metal, decompositions 374
Heating a crystalline solid 49
Hedvall effect,  reaction 356—357 reaction 356—357
Hertz — Knudsen — Langmuir equation 41—42 45
Hertz — Knudsen — Langmuir sublimation model 559—560
Hertz — Knudsen — Langmuir volatilization 307—308
Heterogeneity of reactant crystals 556
Heterogeneous catalysis 47 130
Heterogeneous catalysis and crystolysis 544 560
Heterogeneous catalysis, intermediates 324
Heterogeneous reactions, oxides 294 307—308
Heterogeneous/homogeneous kinetics 527 557 560
Hexaammine chromium(III) compounds, decomposition 504—505
Hexaammine chromium(III) thiocyanate, decomposition 505—506
Hexaammine cobalt(III)compounds, decompositions 495—498 515
Hexaammine copper halides, decompositions 508—509
Hexaammine nickel perchlorate, decomposition 508
Hexacyanoferrates, metal, decompositions 510—511
Hydrate structures 218—219
Hydrates 31 218
Hydride decompositions, reversible 314
Hydrides, decompositions 313—317
Hydrides, surface properties 314
Hydrogen bonding 218
Hydrogen desorption, hydrides 314
Hydrogen evolved during dehydroxylation 282
Hydrogen mobility in hydrides 314
Hydrogen peroxide of crystallization 254
Hydroxides, dehydrations 269—288
Hydroxy halides, metal, decompositions 375
Hydroxy halides, two-metal salts, decompositions 375—376
Hydroxy metal salts, dehydrations 287
Hydroxystannates, metal, decomposition 277
Imperfection interactions 306—307
Imperfection loss and annealing 558
Imperfections 8—15 33 250
Imperfections and electrical conductivity 191—192
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



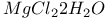 and oxidation of
and oxidation of 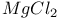
 and
and 
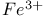 ions in barium azide
ions in barium azide  reaction
reaction