|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Seitz F. — Modern Theory of Solids |
|
|
 |
| Предметный указатель |
 correction 136ff. correction 136ff.
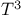 law of specific heats 99 533 law of specific heats 99 533
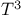 law of specific heats, deviations 120 law of specific heats, deviations 120
Abraham, M. 211 213 637
Absorption coefficient 631ff. 635 646ff.
Absorption coefficient, alkali metals 652
Absorption coefficient, semi-conductors 662
Absorption coefficient, transition metals 656
Absorption spectra, alkali halides 410 446 657ff.
Absorption spectra, F centers 662
Absorption spectra, infrared 665
Absorption spectra, ionic crystals 408ff.
Absorption spectra, metals 651ff.
Absorption spectra, semi-conductors 661ff.
Acceleration in band scheme 315ff.
Accidental degeneracy 290
Activation energy, reactions in solids 474 550
Activation energy, semi-conductors 459ff.
Additivity, atomic heats 38
Additivity, ionic radii 51
Additivity, ionic susceptibilities 59
Additivity, refractivities 660
Adiabatic approximation 470ff.
Ahlberg, J.E. 114ff.
Akulov, N.S. 22ff.
Alkali halides, absorption spectra 408ff. 446
Alkali halides, bands 441
Alkali halides, charge distribution 444
Alkali halides, cohesion 80ff.
Alkali halides, conduction levels 446
Alkali halides, discoloration 460
Alkali halides, F centers 457
Alkali halides, Hall effect 563
Alkali halides, ionic conductivity 55 385ff.
Alkali halides, lattice defects 548ff.
Alkali halides, Madelung constants 78
Alkali halides, optical properties 657ff.
Alkali halides, photoconductivity 413 446 459 563
Alkali halides, semi-conductors 457
Alkali halides, vacancies 458ff.
Alkali metals, Bloch functions 350ff.
Alkali metals, cohesion 348ff. 366
Alkali metals, correlation energy 366
Alkali metals, coulomb field 349
Alkali metals, effective mass 353
Alkali metals, elastic constants 116
Alkali metals, electronic structure 420ff.
Alkali metals, exchange energy 359 421
Alkali metals, ion-core field 348
Alkali metals, level density 366
Alkali metals, optical properties 423 652
Alkali metals, paramagnetism 599
Alkali metals, simple treatment 382
Alkali metals, specific heat 116 421
Alkali metals, total wave function 308ff.
Alkali metals, work function 399
Alkaline earth metals, bands 424ff.
Alkaline earth metals, level density 424
Alkaline earth salts, absorption spectra 408ff.
Alkaline earth salts, cohesion 81ff.
Alkaline earth salts, excitation states 413ff.
Alkaline earth salts, Hall coefficient 467
Alkaline earth salts, photoconductivity 413
Allena J.F. 485
Allison, S.K. 288 475
Allotropy, carbon 484
Allotropy, cobalt 8 487
Allotropy, helium 485
Allotropy, ionic crystals 89ff.
Allotropy, iron 8 487
Allotropy, metals 2
Allotropy, sulfur 484
Allotropy, theory 473ff. 478ff.
Allotropy, tin 8 483
Alloys 25ff.
Alloys, conductivity 541ff.
Alloys, Curie point 45 624
Alloys, diamagnetism 595
Alloys, diffusion 495
Alloys, equilibrium conditions 500
Alloys, exchange integral 624
Alloys, ferromagnetism 45 623ff.
Alloys, filling of levels 434 501
Alloys, heat of formation 38
Alloys, Hume-Rothery rules 28 30ff.
Alloys, interstitial 25ff.
Alloys, magnetic susceptibilities 42ff. 595
Alloys, ordered 37 502
Alloys, phase boundaries 499
Alloys, phase changes 500
Alloys, quenching of magnetization 44ff.
Alloys, resistivity 39ff. 43ff.
Alloys, substitutional 25ff.
Alloys, thermal properties 37
Anderson, C.T. 13 58
Andrews, D.H. 114ff.
Angular momentum, orbital 426 578
Angular momentum, spin 206ff. 232 426 577
Anisotropy 21 627ff.
Antisymmetric states 209 230 213
Antisymmetric states, molecular helium 264
Antisymmetric states, two electrons 232 260
Approximate methods 329ff.
Atomic dipole moment 218
Atomic heat see “Specific heat”
Austin, J.B. 15
Baber, W.G. 640
Baedeker, K. 69 70
Balamuth, L. 96
Band scheme 251 271ff.
Band scheme, alkali halides 441
Band scheme, alkali metals 348 420ff.
Band scheme, anisotropy 628
Band scheme, Boltzmann’s equation 319
Band scheme, conductivity 274
Band scheme, connection with Heitler — London scheme 337
Band scheme, diamond 452
Band scheme, excited states 407 408
Band scheme, ferromagnetism 339
Band scheme, holes 317
Band scheme, ionic crystals 441
Band scheme, level density 307ff.
Band scheme, metals 420ff.
Band scheme, narrow bands 303ff.
Band scheme, overlapping 296
Band scheme, rules concerning 274ff. 295ff.
Band scheme, transition metals 426ff. 468
Banks, F. 495
Bardeen, J. 340 348 352 354 374 381ff. 395 397ff. 400 421ff. 520ff. 530ff.
Barkhausen effect 25
Barkhausen, H. 21
Barnes, R.B. 95 125 666 667
Barnett, S.J. 426
Barrer, R.M. 494
Bartlett, J.H. 251
Bassoe, E. 670
Baumbach, H.H.v. 71
Bearden, J.H. 440
Becker, G. 45ff.
Becker, J.A. 146 162 168 404
Becker, R. 20 211 213 627 637
Beeman, W.W. 440
Beryllium 371ff.
Beryllium atom 247ff.
Beryllium atom, energy 248
Beryllium, level density 371 437
Beryllium, work function 400
Beta brass 35ff. 506
Beta brass, specific heat 37
Bethe, H. 92 140 141 147 166 185 233 314 505 507 531 520 613ff.
| Beyer, J. 556
Bichowsky, F.R. 3 46 72
Biltz, W. 38
Birtwistle, G. 137
Bismuth 425
Bismuth, diamagnetism 595
Bismuth, effective mass 596ff. 599
Bismuth, optical properties 656
Bitter, F. 18 617
Black, M.M. 250 251
Blackman, M. 100 116 117 120 133ff. 597ff. 667
Bleick, W.F. 89 265 269 383 393 493
Bloch functions 272ff.
Bloch functions, accuracy 312
Bloch functions, alkali metals 348 350ff.
Bloch functions, approximate 273 204 303ff. 331ff.
Bloch functions, excitation waves 647ff.
Bloch functions, free electrons 272
Bloch functions, narrow bands see “Approximate methods”
Bloch functions, symmetry 275
Bloch scheme see “Band scheme”
Bloch, F. 140 251 301ff. 303ff. 314ff. 319 516 518 520 530 531ff. 602 617ff. 642 649ff.
Boas, W. 98
Bohr, N. 235 237
Boltzmann, L. 139 143 169 319 479 516ff. 585ff.
Boltzmann’s equation of state 168ff. 173
Boltzmann’s equation of state, in band scheme 319 516ff. 526
Boltzmann’s theorem 99
Boltzmann’s theorem, entire solid 479
Bond functions 210
Borelius, G. 41
Born — Mayer equation 87 96
Born — Mayer equation, justification 445
Born — Oppenheimer equation 470
Born — von Karman boundary conditions 118 121 126 272
Born, M. 76 79 82ff. 85ff. 91 97 99ff. 111 112ff. 117 118 124ff. 138 265 271 272 470 493 553 667
Bose 209
Bottema, J.A. 39
Bouckaert, L.P. 275
Bound electrons 274
Bozorth, R. 21 23 625
Bragg, W.L. 505 507 509 513 544 604 612
Brass system 30 43
Brattain, R.R. 95 125 666 667
Brattain, W.H. 168 404
Breaking strengths 98
Breit, G. 210 643
Bridgman, P.W. 5 137 180 374 381ff.
Briggs, H.B. 652 653ff.
Brillouin functions 581
Brillouin zones 284 287ff. 294ff. 298
Brillouin, L. 100 141 209 234 272 283ff. 384 520 581
Brody, E. 138
Bronsted, J.N. 483 484
Brooks, H. 426 628
Brown, F.W. 246 251 265
Brown, W.F. 20
Bruch-Willstaeter, M. 393ff.
Buehl, R.C. 562 563
Burrau, O. 255ff.
Burton, E.F. 485
Campbell, L.L. 68
Carbon atom 249
Carbon dioxide 394
Carborundum 2 61 63
Carrard, A. 58
Cashman, R.J. 401 670
Cauchy — Poisson relations 94 376
Cellular method 329
Cellular method, alkali metals 349
Cellular method, empty-lattice test 332
Cellular polyhedron 330ff. 352
Channel-Evans, K.M. 28 32
Characteristic temperature, ionic crystals 57 114 134ff.
Characteristic temperature, metals 108 109ff.
Chemical constant 403
Chemical reactions 470
Chodorow, M.I. 275 430ff.
Clark, A.R. 485
Clark, C.W. 15ff. 57 114 117 136 157
Clausius, K. 13 74 485
Closed shells 228 247 262ff. 302ff. 310 346
Closed shells, copper 367
Closed shells, diamagnetism 582
Closed shells, ionic crystals 388
Closed shells, rare gases 393
Closed-shell interaction 262ff.
Closed-shell interaction, alkali metals 360
Closed-shell interaction, metals 376
Cobalt, allotropy 8 487
Cohen, E. 8 483ff.
Coherent scattering 542
Cohesion 345ff.
Cohesion, alkali metals 348 366
Cohesion, alkaline earth salts 81ff.
Cohesion, alloys 38 271ff. 378
Cohesion, carbon dioxide 393
Cohesion, copper 367
Cohesion, Fock approximation 368
Cohesion, ionic crystals 46ff. 78 80ff. 88ff. 271 386ff.
Cohesion, lithium hydride 390ff.
Cohesion, metallic hydrogen 367
Cohesion, metals 3 271ff. 367ff.
Cohesion, molecular solids 73 391ff.
Cohesion, molecules 254ff.
Cohesion, solids 271ff.
Cohesion, transition metals 427ff.
Cohesion, valence crystals 61 271ff.
Collet, P. 19
Collision terms 169 525 538
Competition of energy terms 230
Compressibility 76 138
Compressibility, ionic crystals 391
Compressibility, theory 373ff.
Compton, A.H. 288 475
Condon, E.U. 210 234 246 247 450 458 559 578 643
Conductivity, alkali halides 55 64
Conductivity, alloys 544
Conductivity, band theory 274 .297ff.
Conductivity, change with melting 491
Conductivity, change with order 41 504
Conductivity, classical theory 190
Conductivity, critique of theory 534
Conductivity, cuprous oxide 65
Conductivity, dependence on vapor pressure 70 461
Conductivity, excitation waves 416
Conductivity, ionic solids 55ff.
Conductivity, low temperature 531
Conductivity, magnetic field 184ff.
Conductivity, metals 9 170ff. 174ff. 190 425 516ff.
Conductivity, optical region 631ff. 635ff. 648
Conductivity, semi-conductors 63 189ff. 457 461 465
Conductivity, simple metals 535ff.
Conductivity, surface 642
Conductivity, transition metals 535
Coolidge, A.S. 258ff. 262
Copper, bands 367 423ff.
Copper, cohesion 367
Copper, optical properties 424 654
Copper-gold system 33 36 503
Copper-silver system 33
Correlation energy 231 234
Correlation energy, alkali instate 366
Correlation energy, atoms 247ff.
Correlation energy, beryllium 373
Correlation energy, free electrons 342ff.
Correlation energy, narrow bands 339
Correlation of electrons 231
Correlation of electrons, exchange 240
Correlation of electrons, free electrons 242
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



 correction
correction 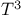 law of specific heats
law of specific heats