|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Bransden B., Joachain C. — Physics of Atoms and Molecules |
|
|
 |
| Предметный указатель |
magnetic quantum number 84 134
Magnetic resonance see Paramagnetic resonance nuclear
Magnetic resonance magneton see Bohr magneton nuclear
Magneton many-electron atoms 290
Many electron atoms, and ionisation potentials 302—7
Many electron atoms, and the central field approximation 292—300
Many electron atoms, and the periodic table 300—7
Many electron atoms, corrections to the central field model 239—52
Many electron atoms, effective potential in 292—5
Many electron atoms, fine structure of 346—52
Many electron atoms, Hartree — Fock model of 320—37
Many electron atoms, hyperfine structure of 371—4
Many electron atoms, Schr dinger equation for 291—2 dinger equation for 291—2
Many electron atoms, the Stark effect in 377—9
Many electron atoms, the Zeeman effect in 374—7
Many electron atoms, Thomas — Fermi model of 313—20
Marsden, E. 23 24 26 40
Maser 164 243 459
Maser, ammonia 567—71
Mass polarisation 250 277 643
Matrix elements of operators 67 69—71
Matrix representation of angular momentum operators 612—14
Matrix representations 69—71
Matrix, diagonal 71
Matrix, equations of motion 72
Matrix, Hermitian, see Hermitian operators
Matrix, unitary 70
Maxwell, J.C. 1 9 155—6
Melvill, Th 27
Mendeleev, D.I. 39 306
Meson 151
Metastable levels 183
Metastable levels, quenching of 224—5
Metastable state 484
Methane molecule 383—4 421 423
Michelson, A. 232
Microwave spectra 386 432
Millikan, R.A. 7 17 18
Millikan’s oil drop experiment 7—8
Minimum principle for the energy 119
Mole 2
Molecular orbitals 405
Molecular orbitals, and atom-atom scattering 543—9
Molecular orbitals, for 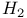 405—8 405—8
Molecular orbitals, for heteronuclear molecules 415—20
Molecular orbitals, for homonuclear molecules 412—14
Molecular orbitals, notation for 413 416
Molecular structure 383
Molecular structure, general nature of 383—6 (see also Diatomic molecule polyatomic
Molecules see Diatomic molecule polyatomic
Moment of inertia of a molecule 385 430
Momentum and the uncertainty principle 57—8
Momentum operator 55
Momentum space wave functions 55 57
Momentum space wave functions, hydrogenic 621—8
Momentum transfer 490 515
Morse potential 391—4
Morse, P.M. 391
Moseley, H. 38 39
Moseley’s law 39
Moseley’s plot 39
Multiplet see Fine structure hyperfine Stark Zeeman
Multiplicities see Spectral terms
Multipole moments 233 (see also Electric dipole moment electric magnetic
Muon 149—50 247
Muonic atoms 150—1 247
Muonium 149
Nacl molecule 418—20
Negative hydrogen ion 249 257—8 260 270 273 275
Neon ground state 335—7
Neutron 26—7
Neutron stars 248
Newton, Sir I. 9 27
NO molecule 397
Noble gases 306
Non-crossing rule 398—9 412
Non-linear optics 572
Non-localised bonds 425—6
Normal modes 421
Normalisation of wave functions 58—60 66
Nuclear magnetic resonance 560—1
Nuclear magneton 234
Nuclear moments 233—4
Nuclear spin 94 233—45 452—5
Nuclear wave equation 388
Nucleus, discovery of 23—6
Nucleus, size of 26
Nucleus, spin of 233—4
Observables 65
Observables, commuting 72—3
Old quantum theory 38 136
One-electron atoms, and expectation values of 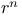 145—7 145—7
One-electron atoms, and parity 144—5
One-electron atoms, and the Stark effect 219—27
One-electron atoms, and the Zeeman effect 207—19
One-electron atoms, energy levels of 133—6
One-electron atoms, field ionisation of 227—9
One-electron atoms, fine structure in 201—7
One-electron atoms, hyperfine structure of 232—45
One-electron atoms, in the Bohr model 29—36
One-electron atoms, photoionisation of 189—93
One-electron atoms, radial distribution function for 142—4
One-electron atoms, Schr dinger equation for 128—33 dinger equation for 128—33
One-electron atoms, spectrum of 133—6 179—80
One-electron atoms, wave functions for 131—3 136—41
One-electron atoms, wave functions in momentum space 621—8
Operators in quantum mechanics 55 61 63 64—5 66—73
Oppenheimer, J. 228
Optical potential 511—13
Optical theorem 468 474 496
Orbital angular momentum 82—4 87—8 614
Orbital angular momentum, commutation relations 82
Orbital angular momentum, eigenfunctions of 84—90 614
Orbital angular momentum, in polar coordinates 83—4
Orbital angular momentum, operators for 82 614
Orbital angular momentum, quantum number 84
Orbital angular momentum, raising and lowering operators 87 614
Orbital angular momentum, spectroscopic notation for 90
Orbitals 141 264 294
Ornstein, Burger and Dorgelo sum rule 369—70
Ortho hydrogen 454
Orthogonality relations, for associated Legendre functions 85
Orthogonality relations, for Legendre polynomials 85 (see also Orthonormality)
Orthonormality 60 66
Orthonormality, for harmonic oscillator wave functions 81
Orthonormality, for spherical harmonics 86 614
Oscillator strengths 181—2
Oscillator strengths, generalised 516—18
Oxygen molecule 383 397 414 438 452—3
P-branch 433—5 441
Pairing 414—15
Para hydrogen 454
Paramagnetic resonance 554—61
Paramagnetic resonance, in solids 559—61
Paramagnetic resonance, Rabi apparatus for 558—9
Paramagnetic resonance, Rabi formula for 558
Paramagnetism 554
Parity 76 82 98—9 144—5 171 178 202 220—2 233 296 358 ungerade
Partial wave cross-section 473
Partial wave expansion 468—75 488
Paschen series 29 205
Paschen — Back effect 214—15 375
Pauli equation 638—9
Pauli exclusion principle, and antisymmetry 106 251 254—5 291 296 299 307 310 406 414 415 452
Pauli spin matrices 94 635
Pauli, W. 233 246 296
Peierls, Sir R.E. 246
Pekeris, C.L. 278 284 285
Periodic system 300—7
Periodic table 39 306—7
| Permutation operator 105—6 250-1
Perot, A. 232
Perrin, J. 3
Perturbation theory, and corrections to the central field approximation 339—41 349—51
Perturbation theory, and correlation energy 339
Perturbation theory, and Raman scattering 436—8
Perturbation theory, for an atom in an electromagnetic field 160—1
Perturbation theory, for fine structure 197—201
Perturbation theory, for scattering see Born approximation
Perturbation theory, of atom-atom interactions 529—32
Perturbation theory, of hyperfine structure 235—40
Perturbation theory, of the Stark effect 219—23 225—7 377-8
Perturbation theory, of the Zeeman effect 214—19 374-7
Perturbation theory, of two-electron atoms 258—71 280-3
Perturbation theory, time-dependent 111—16
Perturbation theory, time-independent 106—11
Pfund series 29
Phase shifts 472—7
Phase shifts, absolute definition of 476
Phase shifts, complex 494—6
Phase shifts, computation of 476
Phase shifts, for a hard sphere potential 479
Phase shifts, integral equation for 476
Phase shifts, near a resonance 480—3
Phosphorescence 446—8
photoelectric effect 15—17 188—93
Photoelectric effect, and Millikan’s experiments 17—18
Photoelectric effect, Einstein’s equation for 17
Photoionisation 188—93
photons 15 17 21 155 157 159
Photons, absorption of 164
Photons, and the Compton effect 20—2
Photons, and the electromagnetic spectrum 18—19
Photons, and the photoelectric effect 17
Photons, emission of 163—66
Photons, parity of 178
Photons, spin of 173—8
Photosphere 584
Pionic atom 152
Planck, M. 12 29
Planck’s constant 13 14 670
Planck’s distribution law 14 169 586
Plane waves 54 100—1 156—7 466 485 504
Polarisability see Dipole polarisability
Polarisation potential 512—13
Polarisation vector 157 173
Polarisation vector, spherical components of 170
Polarisation, circular 174—6
Polarisation, in the Zeeman effect 212—14
Polarisation, linear 157
Polyatomic molecules, electronic structure of 422—6
Polyatomic molecules, rotational motion of 430—1
Polyatomic molecules, vibrational motion of 421
Population inversion 564—6 568
Positron 149 151 633
Positronium 149
Poynting vector 157
Predissociation 444
Pressure broadening 187 588
principal quantum number 133 295
Principal series 362
Pringsheim, E. 10
Probability amplitudes 66 542—6
Probability conservation 62
Probability current density 62 467
probability density 58 141
Proust, J.L. 2
Q-branch 436 441
Quadrupole coupling constant 245
Quantisation of angular momentum 31 44—6
Quantisation of the electromagnetic field 17 155
Quantum defects 360—2
Quantum theory 53 123
Quantum theory, and photons 17 155
Quantum theory, of Planck 12—15
R branch 433—5 441
R ntgen, W.K. 18 ntgen, W.K. 18
Rabi 1 1 558
Rabi flopping formula 558 571
Radial density function 336
Radial quantum number 133
Radial Schr dinger equation, for a free particle 99 dinger equation, for a free particle 99
Radial Schr dinger equation, for central potentials 97 dinger equation, for central potentials 97
Radial Schr dinger equation, for nuclear motion 389 429 dinger equation, for nuclear motion 389 429
Radial Schr dinger equation, for one-electron atoms 129 dinger equation, for one-electron atoms 129
Radial Schr dinger equation, for potential scattering 469 dinger equation, for potential scattering 469
Radial Schr dinger equation, in the Hartree — Fock method 332—5 dinger equation, in the Hartree — Fock method 332—5
Radial wave functions, for a free particle 100
Radial wave functions, for one-electron atoms 136—9
Radial wave functions, for potential scattering 469—73
Radiofrequency spectra in astronomy 583
Rainbow angle 535
Rainbow scattering 538—9
Raman scattering 436—8
Raman, C.V. 436
Rayleigh scattering 436
Rayleigh — Jeans distribution law 12
Rayleigh — Ritz variational method 119
Rayleigh — Schr dinger perturbation theory 106—11 dinger perturbation theory 106—11
Rayleigh, Lord 11 436
Reaction cross section 495
Reactions 462
Reactions, endothermic 463
Reactions, exothermic 463
Recurrence relations for associated Legendre functions 85
Recurrence relations for Hermite polynomials 81
Recurrence relations for Laguerre polynomials 137
Recurrence relations for Legendre polynomials 85
Reduced mass 26 34 102 129 643
Relativistic corrections see Fine structure
Relativistic wave equations 631—41
Representation 69
Resonance lines 36 362—3
Resonances in scattering 287—8 480-4
Retherford, R.C. 207 229
Rigid rotator 90—1 394
Ritz combination principle 29
Ritz, W. 29 361
Robiscoe, R.T. 232
Rotational broadening 588
Rotational constant 391 393 429—36 441-2
Rotational energy see Rotational motion of a molecule
Rotational motion of a molecule 384—6 428—32 448-52
Rotational spectra of diatomic molecules 431—2 448—52
Rotational-vibrational coupling 394
Rovibronic states 389 433
Russell — Saunders coupling 341—9
Russell — Saunders coupling, and selection rules 358
Russell — Saunders coupling, in heavier atoms 368
Russell — Saunders coupling, in two-electron atoms 366
Rutherford model of the atom 26—7
Rutherford scattering 26 493 597—9 607
Rutherford, Lord 23 29
Rydberg atoms 152—3
Rydberg constant 28
Rydberg constant, for deuterium 36
Rydberg constant, for hydrogen 28 36
Rydberg constant, for one-electron atoms 36
Saha, N.M. 586
Satellite astronomy 584
Scalar operators 618
Scattering amplitude 467
Scattering amplitude, for a complex potential 495
Scattering amplitude, for Coulomb scattering 493
Scattering amplitude, integral representation for 488
Scattering amplitude, near a resonance 481—3
Scattering amplitude, partial wave expansion for 473 495
Scattering length 477—8
Scattering of  particles 24—6 particles 24—6
Scattering of atoms by atoms, classification of processes 527—8
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



 dinger equation for
dinger equation for 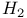
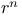
 particles
particles