Авторизация
Поиск по указателям
Prigogine I. — Advances in Chemical Physics, Vol. 2
Обсудите книгу на научном форуме Нашли опечатку?
Название: Advances in Chemical Physics, Vol. 2Автор: Prigogine I. Аннотация: In the last decades, Chemical Physics has attracted an ever increasing amount of interest. The variety of problems, such as those of chemical kinetics, molecular physics, molecular spectros-copy, transport processes, thermodynamics, the study of the state of matter, and the variety of experimental methods used, makes the great development of this field understandable. But the consequence of this breadth of subject matter has been the scattering of the relevant literature in a great number of publications.
Язык: Рубрика: Физика /Статус предметного указателя: Готов указатель с номерами страниц ed2k: ed2k stats Год издания: 1959Количество страниц: 412Добавлена в каталог: 11.04.2010Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
Предметный указатель
158 131 36 37 41 2 6 25 2 29 91 92 117 125 (ref. 145 102 103 195 102 102 195 195 200 193 195 198 195 198 200 102 103 198 195 198 193 199 201 Abkin, A. 151 (ref. 185 Absolute activity 12 13 Absolute intensity 192 Acetaldehyde barrier height of internal rotation 378 382 383 388 Acetonitrile clathrate in hydroquinone 7 11 Acetonitrile, in clathrate 20 Acetyl fluoride height of internal rotation 383 Acetylene hydrate thermodynamic data and lattice constants 8 Acetylene, clathrate in hydroquinone 7 Acrylamides, polymerization of 181 Acrylonitrile 155 Activity coefficients 125 Activity coefficients of copper-gold (Cu 125 Addition polymerization 148 Air 96 107 111 Air 98 Air second virial coefficient 111 Alexander, P. 197 (ref. 205 Alkali metals, electronic correlation energy 252 Aluminum (Al 240 Amino acid 170 Ammonia, clathrates 5 Ammonia, superposition of configurations 296 Anderson, A. R. 35 57 Andrew, S. P. S. 112 115 Anionic polymerization 176 177 181 Anisotropic interaction 33 anthracene 102 153 Antiferromagnetism 313 Antisymmetrical wave functions 260 261 Appel, K. 237 (ref. 322 Arai 235 259 Argon polarizability, magnetic susceptibility and ionization energy 71 Argon polarizability, magnetic susceptibility and ionization energy, potential energy 71 Argon, clathrate 20 21 30 37 41 Argon, hydrate 28 31 32 Argon, hydrate, thermodynamic data and lattice constants 8 Argon, hydrate, vapor pressure 31 Aromatic compound 200 Assayag, P. 129 144 Aston, J. G. 370 371 Atack, D. 103 115 Atoji 73 82 Atom-size effect 141 Attractive intermolecular forces 60 Average potential model 134 Averbach, B. L. 120 (refs. 2 10) 133 58) 144 146 Axilrod 60 82 Axon, H. J. 141 (ref. 144 Baber, T. D. II. 302 Bade, W. L. 60 83 Bahr, K. 151 (ref. 186 Bain, E. C. 123 (ref. 144 Balescu, R. 73 82 Bamford 174 185 band theory 248 Banerjee, S. 200 (ref. 205 Banks, W. P. 9 55 Barrer, R. 15 18 22 23 28 32 55 Barrier forces of internal rotation 381 383 388 391 Barrier values of internal rotation 381 Bartlett, J, H., Jr 297-299 Bawn, C. E. 156 185 Beach, J. Y. 275 (ref. 282 (ref. 322 Beaman, R. G. 155 (ref. 185 Beattie, J. A. 104 115 Beenakker, J. J. 107-109 117 Bellemans, A. 130 (ref. 136 (ref. 146 Benson, S. W. 114 (ref. 115 Benzene photochemical studies 200 Beredjick, N. 169 185 Berg, K. van der 90 (ref. 116 Bergeon, R. 106 117 Beryllium, hydride ion (BeH 324 Beryllium, hydride ion (BeH 299 Beryllium, hydride ion (BeH 324 Beryllium, hydride ion (BeH 249 Beryllium, hydride ion (BeH 302 312 Beryllium, hydride ion (BeH 249 Bethe - Guggenheim pair distribution approximation 122 Bethe Salpeter's Hamiltonian operator 240 Bethe, H. A. 239 319 Bever, M. B. 121 (ref. 146 Bhatt, L. H. 109 118 Bidwell, R. 86 115 Bifunctional monomeric unit 149 Bifunctional polymer 178 Binding mechanism 3 Bird, R. B. 29 56 60 (ref. 70 71 82 106 (ref. 108 116 Bismuth, excess entropy of solution of noble metals in liquid bismuth 133 Bismuth-cadmium alloy (Bi 136 Blankenship, F. F. 9 55 Block polymers 181 Bloembergen, N. 194 205 Blout, E. R. 171 185 Boardman, O. E. 108 110-112 115 Boerboom, A. J. H. 107 108 110 111 116 Bohm, D. 259 Bond energies in the halogens 61 Booth, H. S. 86 115 Born 60 82 Boron 204 Boron fluoride as initiator in polymerization 156 Bouby, L. 197 (ref. 205 Boudreau, R. J. 150 (ref. 186 Boyd, C. A. 110-112 115 Boys, S. F. 257 276 296 Braune, H. 98 115 Braunstein, J. 303 304 Breslow, D. S. 181 (ref. 185 Brickstock, A. 306 Brillouin's theorem 284 Brillouin, L. 283 Brody, H. 177 (ref. 178 (ref. 185 Bromine, hydrate 3 22 43 47 Bromine, hydrate, London energies 78 Bromine, hydrate, molecule hydrate 10 Bromine, hydrate, molecule hydrate, thermodynamic data and lattice constants 71 Bromine, hydrate, molecule, intramolecular London energies 78 Bromoethane, barrier height of internal rotation 382 Bromoethane, barrier height of internal rotation, hydrate 10 Bromoethane, barrier height of internal rotation, hydrate, thermodynamic data and lattice energy 8 Brown, J. F. 163 (ref. 185 Brown, W. B. 183 185 Brownscombe, E. R. 8 41 (ref. 56 Brueckner's approximation 207 273 318 319 323 Brueckner, K. A. 319 Buckingham, R. A. 61 63 64 69 82 Buehler, R. J. 28 (ref. 57 Burkhard, D. G. 378 (ref. 392 Burton 199 200 (ref. 205 Butadiene polymerization 160 Bywater, S. 182 185 Cadmium-lead alloy (Cd 136 Cadmium-magnesium alloy (Cd 136 142 Cadmium-tin alloy (Cd 136 Carbanion 180 Carbon bound 367 Carbon dioxide, clathrate in hydroquinone 7 11 Carbon dioxide, clathrate in hydroquinone, 101 Carbon dioxide, clathrate in hydroquinone, 101 Carbon dioxide, clathrate in hydroquinone, 101 Carbon dioxide, clathrate in hydroquinone, 101 Carbon dioxide, clathrate in hydroquinone, 98 Carbon dioxide, clathrate in hydroquinone, 101 Carbon dioxide, clathrate in hydroquinone, as a “hilfsgas” 18 Carbon dioxide, clathrate in hydroquinone, hydrate 3 Carbon dioxide, clathrate in hydroquinone, hydrate, thermodynamic data and lattice constants 8 Carbon oxide, as radiation source 192 Carbon oxide, as radiation source, gamma rays 203 Carbon tetrachloride-hydrogen sulfide-water ternary system 49 51 52 Carboniuin ion polymerization 158 Carboxylic groups initiator 174 Carmichael, L. 9 42 50 (ref. 57 Carson, D. B. 48 54 55 57 Catalano, E. 61 74 75 82 Catalyst clathrates equilibrium 35 Catterall, J. A. 121 (ref. 132 (ref. 136 (ref. 142 (ref. 144 (ref. 145 Cell partition function, in calculation of thermodynamic quantities of clathrates 26 Cell partition function, in calculation of thermodynamic quantities of clathrates, in calculation of thermodynamic quantities of metallic solutions 135 Cellular method 226 237 252 Ch'en, S. - Y. 114 115 115 Chandrasekar, S. 297 312 Chapiro, A. 197 (ref. 205 Characteristic length and energy 135 Charlesby, A. 197 (ref. 205 Chemical potential of hydrate lattice 22 Cherry, I. J. 297 Chlorine, frequency 189 Chlorine, frequency, hydrate 3 Chlorine, frequency, hydrate, crystal structure of 4 Chlorine, frequency, hydrate, thermodynamic data and lattice constants 8 Chlorine, frequency, London energy 78 Chlorine, frequency, molecule, intramolecular London energy 78 Chlorine, frequency, molecule, intramolecular London energy, magnetic susceptibility of 70 Chlorine, frequency, molecule, intramolecular London energy, polarizability, magnetic susceptibility and ionization energy 71 Chlorine, frequency, polarizability, potential energy 71 Chlorine, frequency, resonance line 190 Chloroethane, barrier height of internal rotation 382 Chloroethane, barrier height of internal rotation, hydrate, thermodynamic data and lattice constants 8 Chloroform, hydrate 3 51 Chloroform, hydrate, hydrate, thermodynamic data and lattice constants 8 Chloroform, hydrate, hydrogen sulfide-water ternary system 49 50 Chloromethane hydrate 50 Chloromethane hydrate, thermodynamic data and lattice constants 8 Chroman complex 4 Clapeyron's equation 23 43 Clathrate compounds 2 4 14 Clathrate compounds, heterogeneous equilibria 5 Clathrate compounds, hydroquinone 38 Clathrate compounds, ideally dilute solutions 17 Clathrate compounds, of hydrogen chlorine 4 Clathrate compounds, solutions 36 Clathrate compounds, stability conditions 5 Claussen, W. F. 4 10 (ref. 55 Cobalt platinum, alloy 128 Cobalt platinum, alloy, enthalpy of formation 144 Cobalt platinum, solid solution 125 Cobalt platinum, solutions 132 Cohen 126 (ref. 133 144 58) 144 Collision diameter 106 Compromise Hartree - Fock function 245 Configurational energy for clathrates 12 Configurations, superposition of 258 Conformal solution theory 137 Coolidge, A. S. 241 251 297 299 304 322 Coordination polymerization 148 162 170 Copeland, C. S. 114 (ref. 115 Copolymerization 168 171 Copper zinc, alloy 129 Copper zinc, solutions 132 Copper, partial molar heat of solution in tin 134 Copper-gold, solid solution (CuAu 129 Copper-gold, solid solution (CuAu 123 Copper-platinum, alloy 128 Copper-platinum, solutions 132 Copper-platinum, superlattice, long range order of 128 Corioles forces 380 Correlated orbitals for different electrons 323 Correlated wave functions 207 223 250 251 257 297 Correlated wave functions, correlation effect in 251 Correlated wave functions, correlation effect in, cancellation of 246 Correlation energy 211 233-236 239 246 254 255 285 Correlation error 224 231-233 242 Correlation factor 257 300 302 303 305 306 311 318 Corte, H. 108 110 111 (ref. 112 117 Cottrell, T. L. 110 111 115 Coulomb, correlation 216 231 232 253 Coulomb, hole 216 218 224 232 305 Coulomb, integral 384 389 Coulson, C. A. 76 82 227 239 241 245 (ref. 257 321 covalent bond 258 Cowley, J. 125 (ref. 127 (ref. 128 144 Craig, D. P. 310 Craig, R. S. 128 (ref. 144 Cramer, F. 5 (refs. 7 8) 55 Critical point, of carbon dioxide 97 Critical point, of the pure substance 96 Critical temperature, for disordering of a superlattice 129 Critical temperature, of air 96 Critical temperature, of ethane 100 Curl, R. F., Jr 371 Curtiss, C. F. 28 (ref. 29 56 57 60 (ref. 70 71 82 106 (ref. 108 116 Cyclic silicones 155 Cyclic silicones, 1-cyclo-hexyl 2,2 didecyl ethane-ethane system 103 Cyclic silicones, monomers 180 Dahler, J. S. 60 82 Dailey, B. 1 188 (ref. 206 377 (ref. 393 Danti, A. 382 (ref. 392 Daudel, R. 258 Dautreppe, D. 191 (refs. 14 15) Davies, P. L. 76 82 Davydov, A. S. 61 82 Dchmelt, H. G. 188 (ref. 205 Dead polymer 173 Dean, C. 192 (ref. 205 Dean, L. B. 389 (ref. 392 Deaton, W. 9 Delocalization of electrons 200 Denbigh, K. G. 80 82 Dennison, D. 370 378 (ref. 381 (ref. 392 Density matrices 207 214 227 231 308 309 314 320 323 Density matrices, variation 319 Depircux, J. 191 (ref. 193 (ref. 194 195 13 20) 196 197 201 (ref. 202 (ref. 205 DeSorbo, W. 132 144 Devonshire, A. F. 24 25 28 (ref. 50 Diamagnetic susceptibility of a spherically symmetrical system 68 Dianin, A. P. 4 55 Dichloroethane dipole moment 372 Dichloromethane thermodynamic data and lattice constants 8 Dichloromonofluoromethane thermodynamic data and lattice constants 8
Реклама
 |
|
О проекте
|
|
О проекте



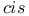 configuration
configuration  -brasses, electronic specific heat of
-brasses, electronic specific heat of 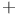 gas clathrate
gas clathrate  -hydroquinone, clathrate compounds of
-hydroquinone, clathrate compounds of 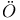 lander, A.
lander, A. 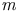 -dinitrobenzene ethylene system
-dinitrobenzene ethylene system 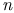 -butane
-butane 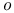 -iodophenol, quadrupole spectrum
-iodophenol, quadrupole spectrum 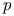 -bromochlorobenzene-ethylene system
-bromochlorobenzene-ethylene system 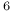 H
H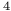
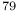 Br), ethylene system
Br), ethylene system 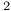
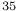 Cl), quadrupole resonance
Cl), quadrupole resonance 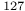 I
I -hexachlorocyclohexane (C
-hexachlorocyclohexane (C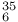 Cl
Cl transitions
transitions 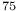 Au
Au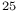 , Cu
, Cu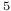 Au
Au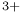 ) electronic correlation energy
) electronic correlation energy 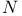 -carboxyanhydride
-carboxyanhydride 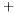 ) electron correlation
) electron correlation 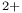 )
) 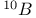 , resonance for the nucleus of
, resonance for the nucleus of 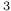 )
)