Авторизация
Поиск по указателям
Carey F.A., Sundberg R.J. — Advanced organic chemistry (Part B)
Обсудите книгу на научном форуме Нашли опечатку?
Название: Advanced organic chemistry (Part B)Авторы: Carey F.A., Sundberg R.J. Аннотация: Since its original appearance in 1977, Advanced Organic Chemistry has found wide use as a text providing broad coverage of the structure, reactivity and synthesis of organic compounds. The Fourth Edition provides updated material but continues the essential elements of the previous edition. The material in Part A is organized on the basis of fundamental structural topics such as structure, stereochemistry, conformation and aromaticity and basic mechanistic types, including nucleophilic substitution, addition reactions, carbonyl chemistry, aromatic substitution and free radical reactions. The material in Part B is organized on the basis of reaction type with emphasis on reactions of importance in laboratory synthesis. As in the earlier editions, the text contains extensive references to both the primary and review literature and provides examples of data and reactions that illustrate and document the generalizations. While the text assumes completion of an introductory course in organic chemistry, it reviews the fundamental concepts for each topic that is discussed. The Fourth Edition updates certain topics that have advanced rapidly in the decade since the Third Edition was published, including computational chemistry, structural manifestations of aromaticity, enantioselective reactions and lanthanide catalysis. The two parts stand alone, although there is considerable cross-referencing. Part A emphasizes quantitative and qualitative description of structural effects on reactivity and mechanism. Part B emphasizes the most general and useful synthetic reactions. The focus is on the core of organic chemistry, but the information provided forms the foundation for future study and research in medicinal and pharmaceutical chemistry, biological chemistry and physical properties of organic compounds.
Язык: Рубрика: Химия /Статус предметного указателя: Готов указатель с номерами страниц ed2k: ed2k stats Издание: 4th editionГод издания: 1938Количество страниц: 965Добавлена в каталог: 16.04.2006Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
Предметный указатель
Halides, aryl, copper-catalyzed coupling 495—498 Halides, aryl, nickel-catalyzed coupling of 527 Halides, aryl, nickel-catalyzed coupling with Grignard reagents 528 Halides, aryl, palladium-catalyzed alkenylation of 503—507 Halides, aryl, palladium-catalyzed coupling with alkenyl stannanes 511—514 Halides, aryl, palladium-catalyzed coupling with alkyl boranes 515—519 Halides, aryl, palladium-catalyzed coupling with arylboronic acids 515—519 Halides, aryl, palladium-catalyzed reactions with organozinc compounds 509 Halides, aryl, preparation from diazonium intermediates 717—721 Halides, reductive dehalogenation of 280 283—284 288—290 296 Halogenation, aromatic 695—699 Halogenation, of acid halides 220 Halogenation, of alkenes 202—205 Halogenation, of alkynes 225—226 Halogenation, of ketones 216—220 Halogenation, reagents for 209—210 Halogenation, stereoselectivity of 201—202 Hard-soft-acid-base theory, application to enolate alkylation 25 Heck reaction 503—507 Heck reaction, intramolecular in Taxol synthesis 885 Hexamethylphosphoric triamide (HMPA), solvation by 21—25 149 280 389 438 HMPA see “Hexamethylphosphoric triamide” Hofmann — Loeffler reaction 655 Hofrnann rearrangement 646—648 Homoenolate anion, synthetic equivalents for 841 Horner — Wittig reaction 117 Hunsdiecker reaction 793 Hydrazones, chiral, enantioselective alkylation of 38 Hydrazones, diazo compounds from 621 Hydrazones, hydrolysis to ketones 38 Hydrazones, N,N-dimethyl, alkylation of anions of 38 Hydrazones, radical addition to 666 Hydrazones, sulfonyl, diazo compounds from 623 Hydrazones, sulfonyl, Shapiro reaction of 309—310 Hydrazones, Wolff — Kishner reduction of 307—308 Hydroboration 226—232 Hydroboration, catalysis of 229—230 Hydroboration, enantioselective 230 236—238 Hydroboration, of 1,5-dienes 871 Hydroboration, of alkynes 239—240 Hydroboration, regioselectivity of 226—227 Hydroboration, stereoselectivity of 227—228 Hydroboration, thermal reversibility of 230—232 Hydroformylation 529—530 Hydrogen atom donors 208—209 290 314 658 664 716 Hydrogen peroxide, in epoxidation 770 Hydrogenation 249—261 Hydrogenation, by dimide 262 Hydrogenation, catalysts for homogeneous 253—259 Hydrogenation, enantioselective 253—259 Hydrogenation, isomerization during 250 Hydrogenation, mechanism of 250 Hydrogenation, stereoselectivity of 252 Hydrogenolysis 260 Hydrosilation 563 567 Hydroxymethylene derivatives, synthesis of 109 Hypohalites, acyl, as halogenating agents 698—699 Hypohalites, ions, in oxidation of methyl ketones 803 Imidazolides see “Acyl imidazolides” Imides, reduction of 99 Imines, addition reactions of 96—100 Imines, anions, alkylation of 31—37 Imines, anions, alkylation of, enantioselective 37—38 Imines, anions, alkylation of, regioselectivity of 37—38 Imines, formation by rearrangement of alkyl nitrenes 644 Imines, reactions with unsaturated silanes 574—575 Imines, reduction by sodium cyanoborohydride 269 Iminium ions, reactions with unsaturated silanes 574 Imino ethers, synthesis of 156 Iodides see “Halides” Iodides, alkenyl, from alkynes via hydroboration 239—240 Iodides, alkyl, preparation from alcohols 146 Iodides, alkyl, reduction by hydride donors 283—284 Iodides, aryl, preparation, by halogenation 688—689 Iodides, aryl, preparation, from diazonium ions 719 721 Iodination, aromatic 688—689 Iodine azide, as reagent 217 Iodine isocyanate, as reagent 217 Iodine nitrate, as reagent 217 Iodine thiocyanate, as reagent 217 Iodolactonization 205—207 Iridium catalysts for homogeneous hydrogenation 253 Isobenzofurans, as Diels — Alder dienes 347 Isoxazoles, from alkenes and nitrile oxides by cycloaddition 365 Isoxazolines, from alkenes and nitrones by cycloaddition 364—365 Jones reagent 748 Julia — Lythgoe alkene synthesis 314 Juvabione, synthesis of 848—859 Ketals, alkenyl, as dienophiles 343—344 Ketals, as protective groups 822—824 829 835—836 Ketenes, as intermediates in diazoketone rearrangements 641—642 Ketenes, dienophilic synthetic equivalent for 341—342 Ketenes, [2 + 2]cycloaddition reactions of 367—369 Ketones, 796 Ketones, 779 Ketones, 298 Ketones, 458 Ketones, 276 Ketones, 391 Ketones, 462 Ketones, 216—218 Ketones, 219 Ketones, 621—622 Ketones, 556 Ketones, 632—633 636—637 Ketones, 641—642 Ketones, 219—220 Ketones, 609—611 Ketones, 779 Ketones, 555—556 Ketones, 462 Ketones, 305—306 779 796—798 800 Ketones, 276—277 Ketones, 487—493 Ketones, 521—523 Ketones, 39—45 89—95 Ketones, 5—10 Ketones, 9—10 26 Ketones, 767 Ketones, 58—60 Ketones, 465 Ketones, 372—374 Ketones, 580 584 Ketones, 453 Ketones, 122—126 Ketones, 11 272—273 292—293 Ketones, 489—490 Ketones, 11 Ketones, 598 Ketones, acylation 108—109 Ketones, Baeyer — Villiger oxidation of 798—800 Ketones, conversion to carboxylic acids by haloform reaction 803 Ketones, enantioselective reduction 278—280 Ketones, enantioselective synthesis of 36—38 493—494 553—554 Ketones, enolates, stereoselective formation 5—10 Ketones, halogenation of 216—219 Ketones, hindered, reaction with organocerium reagents 467 Ketones, hindered, reduction by Grignard reagents 447 Ketones, hindered, Wittig reaction of 112 Ketones, oxidation of 794—803 Ketones, oxidation of, 798 Ketones, oxidation of, Cr(VI) reagents 794—795 Ketones, oxidation of, lead tetraacetate 796 Ketones, oxidation of, N-sulfonyloxaziridines 797—798 Ketones, oxidation of, peroxy acids 798—800 Ketones, oxidation of, selenium dioxide 802 Ketones, photocycloaddition reactions of 372 374—376 Ketones, preparation from, 794 Ketones, preparation from, acid chlorides and Grignard reagents 451 Ketones, preparation from, acid chlorides and organocadmium reagents 464 Ketones, preparation from, acid chlorides and organocopper reagents 485 Ketones, preparation from, acid chlorides and stannanes 525 Ketones, preparation from, alcohols by oxidation 747—757 Ketones, preparation from, alkenes by hydroboration-oxidation 232—235 237—238 Ketones, preparation from, alkenes by palladium-catalyzed oxidation 501 Ketones, preparation from, alkenyl silanes and acid chlorides 568 Ketones, preparation from, alkenyl silanes by epoxidation 780 Ketones, preparation from, alkyl halides by carbonylation 522 Ketones, preparation from, alkynes by hydration 224—225 Ketones, preparation from, aminomethyl carbinols by rearrangement 608 Ketones, preparation from, aromatics by Friedel — Crafts acylation 704—710 Ketones, preparation from, carboxylic acids and organolithium reagents 453—456 Ketones, preparation from, epoxides by Lewis acid-catalyzed rearrangement 778 Ketones, preparation from, nitrites and Grignard reagents 449—450 Ketones, preparation from, organoboranes by carbonylation 550—555 Ketones, protecting groups for 835—837 Ketones, reactions of, with, allylic silanes 568—571 574 Ketones, reactions of, with, azides 650 Ketones, reactions of, with, diazoalkanes 608—609 Ketones, reactions of, with, hydrazoic acid 649 Ketones, reactions of, with, organolithium compounds 453—456 Ketones, reactions of, with, organomagnesium compounds 446—450 457—458 Ketones, reactions of, with, sulfur ylides 122—126 Ketones, reduction by, dissolving metals 292—293 299—305 Ketones, reduction by, Grignard reagents 447 Ketones, reduction by, hydride exchange 287—288 Ketones, reduction by, hydride-donor reagents 262—267 273—280 Ketones, reduction by, silanes 286—287 Ketones, reductive coupling of 299—305 Ketones, reductive deoxygenation of 307—310 Ketones, ring expansion of cyclic 608—609 Ketones, stereoselective reduction of 273—280 Knoevenagel condensation 100—101 Lactams, by iodocyclization of O,N-trimethylsilyl imidates 207 Lactones, 98 Lactones, formation of 170—171 522 636 655 659 801 Lactones, macrocyclic 171—172 Lactones, protection as dithioketals 838 Lactones, reduction of 266 Lanthanide salts, as catalysts, 1,3-dipolar cycloaddition 365 Lanthanide salts, as catalysts, addition reactions of allylic silanes 570 Lanthanide salts, as catalysts, alcohol acylation 167 Lanthanide salts, as catalysts, aromatic nitration 697 Lanthanide salts, as catalysts, Baeyer — Villager reaction 799 Lanthanide salts, as catalysts, conjugate addition 45 Lanthanide salts, as catalysts, Diels — Alder reaction 339 350 Lanthanide salts, as catalysts, ene reactions of aldehydes 401 Lanthanide salts, as catalysts, epoxide ring opening 775 Lanthanide salts, as catalysts, Friedel — Crafts reactions 704 Lanthanide salts, as catalysts, Fries rearrangement 710 Lanthanide salts, as catalysts, hydride transfer 287—288 Lanthanide salts, as catalysts, Mukaiyama reaction 79 Lanthanides, organo- compounds 467—468 Lead tetraacetate, amides, oxidation of 649 Lead tetraacetate, diols, cleavage of 791 Lead tetraacetate, oxidative cyclization of alcohols by 655—656 Lead tetraacetate, oxidative decarboxylation of carboxylic acids 792—794 Lewis acid catalysis for, addition of diazo compounds to ketones 609 Lewis acid catalysis for, addition of silanes to aldehydes 568—573 Lewis acid catalysis for, addition of stannanes to aldehydes 580—585 Lewis acid catalysis for, aromatic halogenation 697 Lewis acid catalysis for, Diels — Alder reactions 336—338 349—350 355 Lewis acid catalysis for, dioxolane formation 835 Lewis acid catalysis for, ene reactions 401—403 Lewis acid catalysis for, Friedel — Crafts reactions 697—711 Lewis acid catalysis for, Mukaiyama reaction 79 82 Lindlar's catalyst 260 Lithium tri-t-butoxyaluminum hydride 267 Lithium trialkylborohydrides, as reducing agents 267 276 278 Lithium triethylborohydride 284 776 Lithium, organo- compounds, alkenyl, alkylation of 445 Lithium, organo- compounds, alkenyl, preparation by Shapiro reaction 444 885 Lithium, organo- compounds, alkylation of 445—446 Lithium, organo- compounds, alkynyl 438 Lithium, organo- compounds, allylic, alkylation of 445 Lithium, organo- compounds, benzylic, alkylation of 445 Lithium, organo- compounds, configurational stability of 442 Lithium, organo- compounds, cyclization of 452 Lithium, organo- compounds, organoboranes from 548 Lithium, organo- compounds, preparation of 436—437 Lithium, organo- compounds, preparation of, by lithiation 438—441 Lithium, organo- compounds, preparation of, from halides by halogen-metal exchange 442—443 Lithium, organo- compounds, preparation of, from hydrazones by Shapiro reaction 444 885 Lithium, organo- compounds, preparation of, from stannanes by metal-metal exchange 443—444 Lithium, organo- compounds, preparation of, from sulfides by reduction 437 Lithium, organo- compounds, reaction with, carbonyl compounds 447—449 457—458 Lithium, organo- compounds, reaction with, carboxylic acids 453 Lithium, organo- compounds, reaction with, halostannanes 579 Lithium, organo- compounds, reaction with, N-methoxy-N-methylamides 456—457 Lithium, organo- compounds, reaction with, trimethylsilyl chloride 563—566 Lithium, organo- compounds, structure of 438—439 Lombardo's reagent 462 Longifolene, synthesis of 859—869 Magnesium, organo- compounds, alkylation of 446 486—487 Magnesium, organo- compounds, alkynyl 438 Magnesium, organo- compounds, copper-catalyzed conjugate addition of 494—497 Magnesium, organo- compounds, cyclopropylmethyl, ring-opening of 452 Magnesium, organo- compounds, mixed copper reagents 495 Magnesium, organo- compounds, nickel-catalyzed coupling 528 Magnesium, organo- compounds, organoboranes from 548 Magnesium, organo- compounds, preparation of 434—435 438 Magnesium, organo- compounds, reactions with, acid chlorides 451 Magnesium, organo- compounds, reactions with, aldehydes 446—450 Magnesium, organo- compounds, reactions with, amides 451 Magnesium, organo- compounds, reactions with, carbon dioxide 451 Magnesium, organo- compounds, reactions with, esters 447—448 Magnesium, organo- compounds, reactions with, halostannanes 579 Magnesium, organo- compounds, reactions with, ketones 446—450 457—458 Magnesium, organo- compounds, reactions with, nitrites 450 Magnesium, organo- compounds, reactions with, triethyl orthoformate 451 Magnesium, organo- compounds, reactions with, trimethylsilyl chloride 563 566 Magnesium, organo- compounds, stereochemistry of 435—436 Magnesium, organo- compounds, structure of 434 436 452 Magnesium, organo- compounds, unsaturated, isomerization of 451—452 Malonate ester anions acylation of 105 108 Malonate ester anions acylation of, alkylation of 11—13 Malonate ester anions acylation of, as enolate synthetic equivalents 13 Malonate ester anions acylation of, cyclization of 13 Malonate ester anions acylation of, reaction with 510 Malonic acids, decarboxylation of 13 Malonic acids, in Knoevenagel condensation 101 Mannich reaction 96—99 Markownikoff's rule 191—192 Meerwein arylation reaction 722 Meerwein — Pondorff — Verley reduction 287 Mercurinium ion intermediate 196 Mercury compounds, organo- 464—465 Mercury compounds, organo-, 659 Mercury compounds, organo-, aromatic 711—713 Mercury compounds, organo-, carbenes from 625 633 Mercury compounds, organo-, preparation of 196—200 464—465 659 Mercury compounds, organo-, reactions of 465 Mercury compounds, organo-, reduction by sodium borohydride 196—198 659 Mercury salts in, aromatic halogenation 698 Mercury salts in, aromatic mercuration 711—713 Mercury salts in, initiation of polyene cyclization by 600 Mercury salts in, oxymercuration reactions 196—200 Mercury salts in, ring-opening of cyclopropanes by 856 Michael reaction see “Conjugate addition” Michaelis — Arbuzov reaction 158 Mitsunobu reaction, in nitrogen alkylation 157 Mitsunobu reaction, in preparation of alkyl azides 151 Mitsunobu reaction, in preparation of alkyl iodides 146 Mitsunobu reaction, inversion of alcohol configuration by 153—154 Mukaiyama reaction 78—82 Mukaiyama reaction, in synthesis of Taxol 887 Mukaiyama reaction, intramolecular in synthesis of longifolene 868 N-bromosuccinimide, aromatic bromination 697 N-bromosuccinimide, bromination of ketones by 216—219 N-bromosuccinimide, bromohydrins from alkenes by 203
Реклама
 |
|
О проекте
|
|
О проекте



 -acetoxy, by oxidation with lead tetraacetate
-acetoxy, by oxidation with lead tetraacetate  -unsaturated, addition of organocopper reagents to
-unsaturated, addition of organocopper reagents to 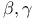 -unsaturated by alkene arylation
-unsaturated by alkene arylation  pyridine HMPA
pyridine HMPA 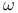 -haloalkyl
-haloalkyl  -allylpalladium compounds
-allylpalladium compounds