|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Lambert J.B., Mazzola E.P. — Nuclear Magnetic Resonance Spectroscopy |
|
|
 |
| Предметный указатель |
Data points, for 1D spectral description (np/2) 49—52
Data points, for first (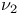 ) dimension spectral acquisition 241 247 251 ) dimension spectral acquisition 241 247 251
Data points, for second (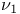 ) dimension spectral acquisition 247 251 ) dimension spectral acquisition 247 251
Data points, number (np) for 1D spectral acquisition 41 49—51
Data sets, imaginary 247
Data sets, real 247
Data-processing parameters 243—250
DAVINS 116 314
Decalins 124
Decane, carbon relaxation 135
Deceptive simplicity 116—117 315
Decoupler, calibration 60—61
Decoupler, field strength 47 60
Decoupler, modulation frequency 58 61
Decoupler, offset 47
Decoupler, sample heating effects 243 258 260
Decoupler, status 47
Decoupling 18 25 46—47 80 105 136 144—146 150 152 155 158—160 189 see "Heteronuclear "Double
Degassing 34
Delay time 132
Dephasing 203—204
DEPT 156—157 161—162 204 207 220 221—222 225—226 229—231 236—237 280—282
Depth gauge 34
Deuterated solvents for locking 33 35
Deuterated solvents, chemical shifts 55
Diamagnetic anisotropy 64—69 71—74 81 86
Diamagnetic anisotropy, shielding 62 68 80
Diamagnetic anisotropy, susceptibility 65—66
Diamagnetic anisotropy, susceptibility, bulk 77
Diastereotopic groups 103—104 138 140 146 179—180 182 189 193 261—262 279 334—335 339—341
Dichloroethene, HCC coupling 108
Dichlorophenols,  spectra 125—126 spectra 125—126
dielectric constant 77—78
Dienes, vicinal couplings 112
Diethyl ether,  spectrum 16 spectrum 16
Difference decoupling 145
Difference NOE 151
Difference spectra 237 see
Difluoromethane 99—100
Digital resolution, in 1D spectra 50—52 180
Digital resolution, in the first (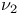 ) dimension in 2D spectra 246—247 ) dimension in 2D spectra 246—247
Digital resolution, in the second (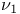 ) dimension in 2D spectra 246—247 ) dimension in 2D spectra 246—247
Digital resolution, optimizing 247
Digital signal filtration 46
Dihedral angle 283 285—286 291 293
Dimensionless spin 295 298
Dimethyl sulfoxide, nonexchanging solvent 75
Diphenylketimine, 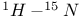 coupling constant 106 coupling constant 106
Dipolar decoupling 25 27
Dipole-dipole coupling 25—26 267
Dipole-dipole coupling, relaxation 131—132 150 152 195—196 198 205 317—321 331—332
DIPSI-2 256 see
Dirac notation 303
Direct coupling (D) 105
Dispersion mode 53 183—184 247 300—301 330 see
Dispersive tails, in phase-sensitive COSY spectra 252
Dispersive tails, with absolute-value data 246 see phase-twisted"
Disulfides, hindered rotation 138
DNMR3 142
Double bond, shielding 67—68
Double irradiation 26 see
Double quantum coherence 162—163 165 183 189 199 204 326—332
Double quantum filtration 183—185
Double quantum transition 304
Double resonance 18 131 136 143—155 160
Doublet of doublets 20
DPFGSE 205
DPFGSE-NOE 240 268 see
DQF-COSY 183—184 207 214—216 221 246 253—257 332
Dreiding model 291
Dummy scans see "Steady-state scans"
Dwell time 40
Dynamic effects 23—25
Dynamic effects, NMR 136—143
EBURP 166
Echo experiment 331
Effective correlation time 132 135 198 317—319
Effective correlation time, nuclear charge 106
Effective correlation time, spin-lattice relaxation time 233
Effective correlation time, spin-spin relaxation time 241 248
Electric-field effect 77—79
Electric-field effect, shielding 69
Electromagnets 31 36—37
Electron density 80
Electron density, spin relaxation 321
Electron spin resonance 183
Electronegativity 6 62—63 81
Electronegativity and vicinal couplings 111 see
Empirical correlations, for  chemical shifts 82—88 92 chemical shifts 82—88 92
Empirical correlations, for  chemical shifts 71—76 91 chemical shifts 71—76 91
Enantiotopic groups 99—100 103 279 334—335 337—341
Energy-level diagram 304—310
Enol ethers,  chemical shifts 86 chemical shifts 86
Epoxides, vicinal coupling 111
Equatorial position 66—67 83—84 110 291—292
Equatorial position, protons, relaxation 134
Ernst angle 43
Esters,  chemical shifts 88 chemical shifts 88
Esters,  chemical shifts 70 chemical shifts 70
Ethane,  chemical shift 81—82 chemical shift 81—82
Ethane, 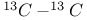 coupling constant 106 coupling constant 106
Ethane,  chemical shift 63 66 69—70 chemical shift 63 66 69—70
Ethane, HCC coupling 108
Ethanol 68
Ethene,  chemical shift 81 chemical shift 81
Ethene, 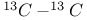 coupling constant 106 coupling constant 106
Ethene,  chemical shift 63—64 66 71 chemical shift 63—64 66 71
Ethene, 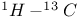 coupling constant 105 coupling constant 105
Ethene, HCC coupling 108
Ethene, HCH coupling 107
Ethers,  chemical shifts 84 chemical shifts 84
Ethers,  chemical shifts 70 chemical shifts 70
Ethyl trans-crotonate 144
Ethyl trans-crotonate,  chemical shift 63 chemical shift 63
Ethyl trans-crotonate,  spectrum 19—20 spectrum 19—20
Ethylbenzene 102—103
Ethyne,  chemical shift 81 chemical shift 81
Ethyne, 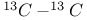 coupling constant 106 coupling constant 106
Ethyne,  chemical shift 63 66 71 chemical shift 63 66 71
Ethyne, 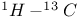 coupling constant 105 coupling constant 105
Ethyne, HCC coupling 108
Evolution period 173 192
EXAN II 318
Exchangeable protons 75
Excitation sculpting 205
Exponential weighting 48—49
EXSY 196 198—199 207 232 268—269 331—332
EXSY, phase-sensitive 199
Extreme narrowing condition 318—321
Extreme narrowing condition, limit 132 149—151 197
F,F-COSY 187
False peaks 180
Fast exchange 23—24 137 143 198
FDM see "Filter diagonalization method"
Fermi contact mechanism 104—105
FID see "Free induction decay"
Field sweep 7
Filter bandwidth 42 57
Filter diagonalization method 250
Filtering of sample solutions 33—34
Filters, analog 42 46
Filters, digital 46
First-order spectra 13 56 98—99 101—102 115 123 125 145 303—305
First-order spectra, pseudo 56
Flip angle, 1D 43—45
Flip angle, 2D 242 see
Flip-flop mechanism 134
Flock 192—193 264—266
FLOCK, BIRD pulse functions 265—266
Fluorine couplings 109 114—115 119
| Fluoromethane 5
Fluxional molecules 140—142
Folding 180 see
Forbidden transitions 304
Formaldehyde, HCH coupling 107
Four-dimensional NMR 203
Fourier analysis 12
Fourier transformation 13 49 172 174 183 201 330
Fourier transformation, number 50 56
Fourier transformation, spectrometer operation 31—32
Free-induction decay 11—13 36 172 174 187
Frequency domain 13
ft see "Fourier transformation"
Full width at half maximum 52 see
FWHM see "Full width at half maximum"
Gamma effect, on  chemical shifts 82—85 chemical shifts 82—85
GARP 258—259 see
Gated decoupling 48 151—152 158
Geminal coupling 17 105—109 120 123 176 182 189 207 315
Gentamycin, DEPT spectra 156—157
Glycine 177—178 180
Gradient coils 39
Gradient coils, echo 204—205
Gradient coils, pulse 203 see
Gyromagnetic ratio ( ) 2—4 22 106—107 132 146 149—150 153 159 193 295 319 321 ) 2—4 22 106—107 132 146 149—150 153 159 193 295 319 321
Half-chair conformation 291
Haloalkanes,  chemical shifts 80—81 84—85 chemical shifts 80—81 84—85
Haloalkanes,  chemical shifts 62—63 68 71 chemical shifts 62—63 68 71
Haloalkanes, hindered rotation 138
Halogenated alkanes 138
Hamiltonian matrix 304—305 307—309
Hamiltonian matrix, operator 302—304 322—324
Hard pulses 165
Hartmann — Hahn condition 27 185
Heavy atom effect 81
Heisenberg uncertainty principle 22
Heptamethylbenzenium ion 198
HETCOR 187—189 193—195 203 207 216—217 220 222—232 261—262
Heterocycles, ring reversal 139
Heteronuclear correlation 187—195
Heteronuclear correlation, decoupling 146—148 151 164—165
Heteronuclear correlation, multiple quantum coherence 161
Heterotopic groups 340
Hindered rotation 137—139
HMBC 193—194 207 262—264 284
HMBC, 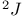 , , 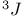 -HMBC modification 274—276 -HMBC modification 274—276
HMBC, gradient version 263—264
HMBC, nongradient version 263—264
HMQC 189—190 193 201—202 204 207 258—259
HMQC, gradient version 258
HOHAHA 185 198
Homoallylic coupling 113 282 287
Homogeneity see "Magnetic-field homogeneity"
Homomorphic groups 340
Homonuclear decoupling 146 151
Homotopic groups 99—100 335—337
HSQC 192—193 258—261 281—282
HSQC, gradient version 259—261
HSQC, multiplicity-edited 271—273
HSQC-TOCSY 266—267
hybridization 63 66—68
Hybridization and coupling 105—106
Hydride shifts 141
Hydrogen bonding 78
Hydrogen-bonding effects, on  chemical shift 68 75 chemical shift 68 75
Hydroxyl protons 75
Hydroxyl protons, resonance 23
Hypercomplex Fourier transform 241
Hyperconjugation 107 113
Imaginary part 49 183 299—300
Imaginary part, spectrum 50 see
Imines, HCN coupling 108
INADEQUATE 106 162—163 165 183 189 204
INADEQUATE, 2D 199—200 207 213 232 332—333
Increments see "Time increments"
Indene, allylic coupling 113
Inductive effect 63 68—70 72—74 80 84—85 112 182 193 see
INEPT 156—162 187 192 325
INEPT-INADEQUATE 200 207
Inhomogeneity effects 37—38
Integration 19 21 57—58
Interleaved acquisition 237 243 see
Inverse detection 189—190
Inverse-gated decoupling 48
Inversion recovery 133 135 163—165 168 234 327—328
Isobutane,  chemical shifts 83 chemical shifts 83
Isobutylene,  chemical shift 86 chemical shift 86
Isobutylene,  chemical shift 71 chemical shift 71
Isochronous 102 334—336 338
Isomontanolide 213
Isonitrile 167
Isopropyl group 17 103
Isotope effects, on chemical shifts 79 118
Isotropic groups 63—64
Isotropic mixing sequences, DIPSI-2 256
Isotropic mixing sequences, GARP 258—259
Isotropic mixing sequences, MLEV-17 256
Isotropic mixing sequences, WALTZ-16 256 258 262 266
Isotropic mixing sequences, WURST 258—259
J-filter, in HMBC experiment 262
J-resolved spectroscopy 186—187 207
Jeener experiment 172 176
Karplus equation 76 109—112 193 285 324
Ketones,  chemical shifts 88 chemical shifts 88
Laboratory time scale 136
Lactones,  chemical shifts 88 chemical shifts 88
Lanthanide shift reagents 116
LAOCN 3 116 314
Larmor frequency 3—4 8—10 12 131 143 148 162 187 190 203 296 317—318 321 323—324 328 333
Leucine 177—180
Line broadening 48 238 240
Line broadening, functions 48—49
Line broadening, parameter 58—61
Line shape 36
Line shape, absorption mode 52
Line shape, phase-twisted 246 252
line width 10 12 302
Line width at half-height see "Full width at half maximum"
Linear prediction 247—250
Linear prediction, backward 250
Linear prediction, extensions 248 251 258 260
Liquid crystal solvent 105
Local electron currents 62—63
Lock, external 35
Lock, internal 35
Lock, optimizing 36
Lock, phase 35—36
Lock, receiver 35
Lock, saturation 35 37 237—238
Lock, signal 33 35—36
Lock, transmitter 35
Locking, automatic 35
Locking, field/frequency 35—36
Lone pair, shielding 63—68
Long-range COSY 182—183 207 253
Long-range COSY, coupling 105 112—115 124 177 182—183 192—193 207
Longitudinal relaxation see "Spin-lattice relaxation"
Lowering operator 306
Lutidines,  spectra 124—125 spectra 124—125
Lysine 184—185
Magic angle 65
Magic-angle spinning 26 32
Magnetic equivalence 99—104 106 115 117 122 334 336—339
Magnetic equivalence, moment 1 3 5 14 131 144 295 298 302 321
Magnetic equivalence, resonance imaging 203
Magnetic-field homogeneity 36 52
Magnetic-field homogeneity, optimizing 37
magnetization 8—12 131 134 158—159 161 163 172—173 175 180 184—185 187 189—190 192 195—197 203—204 297—301 322—331
Magnetization, transfer 143 146 175—176 181—183 185 187 196—199 see
Magnetogyric ratio 2 see
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



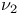 ) dimension spectral acquisition
) dimension spectral acquisition 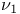 ) dimension spectral acquisition
) dimension spectral acquisition  spectra
spectra 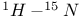 coupling constant
coupling constant  chemical shifts
chemical shifts 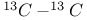 coupling constant
coupling constant 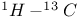 coupling constant
coupling constant  )
) 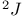 ,
, 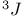 -HMBC modification
-HMBC modification