|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Fowler R.H. — Statistical thermodynamics: A version of statistical mechanics for students of physics and chemistry |

Обсудите книгу на
Нашли опечатку?
Выделите ее мышкой и нажмите Ctrl+Enter
|
Название: Statistical thermodynamics: A version of statistical mechanics for students of physics and chemistry
Автор: Fowler R.H.
Язык: 
Рубрика: Физика/
Статус предметного указателя: Готов указатель с номерами страниц
ed2k:
Год издания: 1956
Количество страниц: 701
Добавлена в каталог: 26.03.2008
Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
|
|
 |
| Предметный указатель |
External reactions of assembly 54
Eyring and Hirschfelder free volume of liquids 332
Eyring and others refined theory of reaction rates 514—517
Eyring entropy of liquids 327
Fermi — Dirac statistics 47 49 159
Fermi — Dirac statistics for electron gas 453
Ferromagnetism, collective electron, Stoner’s treatment 678—685
Ferromagnetism, Curie temperature for 677
Ferromagnetism, Heisenberg’s explanation of 677
Ferromagnetism, ideal, laws for 674
Ferromagnetism, Weiss’s theory of 675—677
Free energy in magnetic field 673
Free energy of adhesion between liquid and vapour 445—448
Free energy of electrolytes, Debye’s approximation 392
Free energy of liquid, form of 322—324
Free energy of perfect gas 71
Free energy, configurational for non-equivalent sublattices 598—604
Free energy, configurational, power series for 574—576
Free energy, defined 58
Free volume of liquid 331—334
Fugacity, defined 266
Fundamental formula, defined 59
Fuoss’s treatment of ionic association 413—415
Gases, mixtures of, entropy of 162—164
Gases, mixtures of, free energy of 162—164
Gases, refractive index for 641—643
Giauque and others vapour pressure data 203
Giauque, comparison of calorimetric and spectroscopic entropy 196 210—215
Giauque, definition of ice point 69
Gibbs adsorption formula 423 444
Gibbs chemical potential 301
Gibbs condition for dissociative equilibrium 162
Gibbs definition of partial potentials 59
Gibbs elementary principles in statistical mechanics 55
Gibbs function 233
Gibbs grand canonical ensemble 231
Gibbs — Duhem relation 233 422
Gillespie and others solubility of H in Pd 561
Glasses, entropy of 217—219
Gorsky approximation for order 574
Grand canonical ensemble of Gibbs 231
Grand partition function 231 sqq.
Grand partition function for crystals 240—242
Grand partition function for crystals, general imperfect 552
Grand partition function for crystals, perfect mixed 242—244
Grand partition function for electron gas 667 681
Grand partition function for gases 236—239
Grand partition function for groups of sites 589—595
Grand Partition function for regular assemblies 244—253
Grand Partition function for surface phase, gaseous 239
Grand partition function, constructed and used 234
Gronwall and others solution of Poisson — Boltzmann equation 408
Guentelberg activity coefficients, ionic 407
Hamiltonian energy for rigid body 106
Hamiltonian, classical for systems in external field 608—610
Hamilton’s equations of motion 8
harmonic oscillator 25—28
Harmonic oscillator, partition function for 40—42
Heat capacity of crystals 141—149
Heat capacity of crystals Debye’s approximation 141—144
Heat capacity of crystals, atomic, 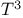 -law for 142 -law for 142
Heat capacity of crystals, molecular, 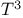 -law for 153 -law for 153
Heat capacity of hydrogen gas at high temperatures 98
Heat capacity of perfect gases 80 sqq.
Heat capacity of perfect gases, diatomic 90
Heat capacity of perfect gases, polyatomic 111—121
Heat capacity via partition functions 68
Heat capacity, anomalous configurational 583—586
Heat capacity, rotational, for 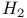 at low temperatures 91—94 at low temperatures 91—94
Heat capacity, vibrational slow attainment of equilibrium 100—102
Heat capacity, vibrational, for diatomic molecules at high temperatures 94—102
Heat of dilution, of electrolytes 403—405
Heat of dilutions, of solutions, dilute 374
Heat of evaporation of liquids 349
Heitler and London overlap energy 278
Helmholtz free energy 233
Henry heat capacity of simple gases 101
Herzfeld diameters of molecules 277
Heterogeneous reactions, equilibrium of 183
Hildebrand regular solutions, defined 245
Hildebrand rule 350
Hildebrand solutions of molecules of different lengths 370
Hinsheiwood unimolecular reactions 528
Hirschfelder and others entropy of melting 330
Hirschfelder and others equation of state at high pressures 289
Hirschfelder and others Joule — Thomson coefficients 288
Homogeneous reactions in Solutions, dilute 375
Homogeneous reactions, equilibrium of 156 sqq.
Hovorka and Rodebush osmotic coefficients 402
Hueckel electrolytes, theory of 394
Hund paramagnetic susceptibility 619
Hydration in electrolytes, effects of 379
Hydration in electrolytes, general discussion of 379—381
Ice point, Giauque’s definition of 69
Ideal assemblies, defined 244
Ieotopic diatomic molecules, dissociation of 167—169
Impacts on a plane, number of, in perfect gas 124
Imperfect crystals 541 sqq.
Imperfect crystals of binary salts 545—549
Imperfect crystals of binary salts with excess of one component 549—552
Imperfect crystals, activation energy for transport in 549
Imperfect crystals, general, grand partition function for 552
Imperfect crystals, simple pure 542—545
Imperfect gases 255 sqq.
Imperfect gases at high pressures 289—291
Imperfect gases in external fields 300
Imperfect gases, chemical equilibrium in 271
Imperfect gases, dielectric constant of 650
Imperfect gases, distribution laws in 259—261
Imperfect gases, empirical equations of state for 273—276
Imperfect gases, general theory of 301 sqq.
Imperfect gases, partition function for, first approximation 262—265
Imperfect gases, partition function for, for potential energy 256—259
Imperfect gases, refractive index of 650
Imperfect gases, simple models 272
Imperfect gases, thermodynamic functions for 256—259 265—267
Impurities, solutions of, in crystals 552
Insulators and metals, compared 465—469
Intermolecular energy 276 sqq.
Intermolecular energy, analysis of 291—296
Internal rotations in polyatomic molecules 107—109
Ionic association, Bjerrum’s theory of 409—412
Ionic association, Fuoss’s treatment of 413—415
Ionic atmosphere, mean thickness of 393
Ionic diameter, effect of, in Debye’s approximation 397
Ionic interaction, specific 415—420
Ionic strength of electrolyte, defined 393
Isomeric equilibrium 157
Isotherms for 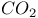 , observed 302 , observed 302
Isotherms Langmuir’s adsorption 427
Isotherms near critical temperature 314—318
Isotherms pressure-composition, for H in Pd 560—563
Isotherms, metastable 304 314
Isotopic mixed crystals, vapour pressure of 186
Isotopic mixtures, chemical constants of 174
Jahn and Teller theorem on crystalline fields 628
Joule — Thomson coefficient 266 288
Joule — Thomson coefficient for binary mixtures 299
Joule — Thomson coefficient, inversion temperature for 267
Kahn equation of state of gas 256 301
Kamerlingh Onnes and Keesom equations of state of gases 273
Keesom equation of state of gas 273
Keesom second virial coefficient for polar molecule 294
Kelvin temperature scale 39
Kinetic salt effects in bimolecular reaction rates 536—540
Kirkwood configurational free energy 574
Kirkwood critique of Debye’s theory 406
Kramers crystalline fields, theorem on 627
Kramers electrolytes at high dilution 406
Lacher adsorption isotherm, refined theory 442
| Lacher solubility of H in Pd 558
Landau electron diamagnetism 669
Lange and Robinson heat of dilution of electrolytes 404
Langevin function 630 676
Langevin paramagnetic susceptibility, theory of 620
Langmuir adsorption isotherm 422 427
Larmor’s theorem 615
Lennard — Jonee and Devonshire free volume of liquids 332
Lennard — Jonee and Devonshire method of evaluating 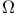 for liquids 323 for liquids 323
Lennard — Jones second virial coefficient 280
Lewis and Gibson third law of thermodynamics 224
Lewis fugacity, defined 266
Limiting principle 9 25 26—29
Lindemann unimolecular reactions 519
Liquids 319 sqq.
Liquids and vapour interface, surface energy of 448—451
Liquids and vapour, equilibrium of, refined model 346—349
Liquids relationship to gases and crystals 326
Liquids, associated, defined 320
Liquids, crude models of 324—326
Liquids, crude models of, thermodynamic functions for 328
Liquids, dielectric constant of 217—219 (see dielectric constant entropy of)
Liquids, free volume of 331—334
Liquids, harmonic oscillator model for 325
Liquids, heat of evaporation of 349
Liquids, normal, defined 319
Liquids, refined model of 336—350
Liquids, refined model of, corresponding states for 342—345
Liquids, refined model of, thermodynamic functions for 342—345
Liquids, refractive index for 641—643
Liquids, smoothed potential model for 325
Localized assemblies, complexions enumerated for 29
London dispersion energy 278
London second virial coefficient 296
Lorenz — Lorentz formula for refractive index 642
Macleod equation for surface tension 450
Magnetic field, free energy in 673
Magnetic interactions, effect of, on thermodynamic functions 658—660
magnetic susceptibility 608 sqq. (see also diamagnetic s. paramagnetic
Magnetic susceptibility of alkali metals 670
Magnetic susceptibility of metals 666—671
Magnetic susceptibility, atomic, defined 613
Magneton number 621
Matter, atomic constitution of 3
Maxwell’s law 73—77
Mayer equation of state of gas 276 301
Mechanism of gas reactions, bimolecular, defined 500
Mechanism of gas reactions, unimolecular, defined 500
Melting, elementary theory of 329—331
Melting, entropy of 329
Metallic alloys, order-disorder in 563 sqq.
Metals and insulators, compared 465—469
Metals, electron theory of 452 sqq.
Metals, magnetic susceptibility of 666—671
Metals, solutions of H in 554—563
Mixing, entropy and free energy of, in gases 163
Molecular energy, defined 72
Monolayer, ideal localized 426—428
Monolayer, ideal localized, spreading pressure for 443
Monolayer, mobile 423—426
Monolayer, regular localized 429—443
Monolayer, regular localized, crude approximation 430—433
Monolayer, regular localized, grand partition function for 438—441
Monolayer, regular localized, refined treatment of 437—443
Monolayer, regular localized, two-phase 433—435
Nernst’s formula for equilibrium constant 173
Nernst’s heat theorem 92 184 191 223
Nernst’s heat theorem, conditions for validity of 226—229
Nernst’s heat theorem, deduced from third law 226
Nernst’s identification of vapour pressure and chemical constants 183
Nernst’s principle of unattainability of absolute zero 224
Non-localized assemblies, complexions enumerated for 30
Normal modes of crystals 130—134
Normal modes of crystals, Bora’s analysis of 152
Normal modes of crystals, Debye terms 151
Normal modes of crystals, Einstein terms 151
Normal modes of elastic continuum 127
Nuclear spin 84—87
Nuclear spin in atomic partition functions 87
Nuclear symmetry in diatomic molecules 84—87
Onsager critique of Debye’s theory of electrolytes 405
Order, long range, defined 567
Order-disorder comparison of approximations 581
Order-disorder comparison of theory and experiment 586—589
Order-disorder in metallic alloys 563 sqq.
Order-disorder quasi-chemical method 576—581
Order-disorder, Curie temperature for 573
Order-disorder, Curie temperature for, variation with composition 589
Order-disorder, variations of type of 605
Order-disorder, Zeroth approximation for 570—574
Order—disorder, Zeroth approximation for, for 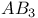 lattices 600—603 lattices 600—603
Orientational degeneracy frozen in 220—223
Ortho-molecules, defined 91
Ortho-para separations, effect of, on equilibrium 187
Oscillators, localized, assemblies of 30—34
Osmotic coefficients from Debye’s approximation 399—401
Osmotic coefficients in electrolytes 381—383
Osmotic coefficients, compared with experiment 401—403
Overlap energy 277
Para-molecules, defined 91
Paramagnetic salts below 1° K. 661
Paramagnetic salts, entropy of 652
Paramagnetic saturation 629
Paramagnetic susceptibility in crystalline field 626—629
Paramagnetic susceptibility of atoms 618
Paramagnetic susceptibility of electron gas 667
Paramagnetic susceptibility of free molecules 631—633
Paramagnetic susceptibility of rare earth salts 621—624
Paramagnetic susceptibility of transition element salts 624
Paramagnetic susceptibility, Curie’s law for 619
Paramagnetic susceptibility, defined 610
Paramagnetic susceptibility, Hund’s formula for 619
Paramagnetic susceptibility, Langevin’s formula for 620
Paramagnetic susceptibility, Weise’s law for 627
Partial molecular volume, defined 72
Partial potential of crystal, final proof 178
Partial potential, defined 59
Partial pressure of perfect gas 71
Partition function for configurations of given order 568
Partition function for continuum 129
Partition function for crystals, Debye’s approximation 133
Partition function for crystals, electronic factors for 134
Partition function for crystals, nuclear factors for 134
Partition function for crystals, perfect mixed 154
Partition function for electrolytes, dimensional analysis 384
Partition function for electrolytes, effect of electrostatic forces on 383
Partition function for electrolytes, Milner’s formula 384
Partition function for electron spins in crystalline field 657
Partition function for electronic degrees of freedom 79 102—106
Partition function for harmonic oscillators 40—42
Partition function for imperfect gases first approximation 262—265
Partition function for imperfect gases, evaluated by association theory 267—270
Partition function for imperfect gases, structure of, general theory 301 sqq.
Partition function for perfect gases see perfect gases
Partition function for rigid rotators 43—45 106
Partition function for rigid rotators with electric dipoles 633 635
Partition function for rigid rotators with magnetic dipoles 620 629 652
Partition function for structureless particles in a box 52—54
Partition function for systems of several freedoms 39
Partition function for systems of several freedoms in external fields 612
Partition function for temperature radiation 129
Partition function, classical, for rigid body 106
Partition function, defined 33
Partition function, grand, further generalization of 253
Partition function, rotational, for 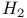 at low temperatures 91—94 at low temperatures 91—94
Pauli exclusion principle 17
Pauli theory of electron paramagnetism 667
Pauling entropy of ice 214
Pauling rotations in solids 151
Peierls order in non-equivalent sublattices 603
|
|
 |
| Реклама |
 |
|
|
 |
 |
|
 |
|
О проекте
|
|
О проекте



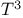 -law for
-law for 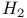 at low temperatures
at low temperatures 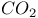 , observed
, observed 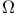 for liquids
for liquids 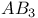 lattices
lattices