|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Flogge S. (ed.) — Encyclopedia of Physics. Thermodynamics of Gases |
|
|
 |
| Предметный указатель |
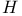 -function -function 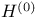 241 241
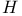 -function -function 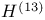 275 275
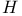 -function -function 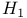 239 239
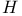 -function -function 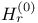 241 241
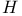 -function -function 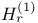 241 241
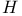 -function -function 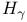 213 225 226 228 213 225 226 228
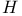 -function -function 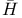 239 239
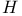 -Theorem 239 241 358 484 489 -Theorem 239 241 358 484 489
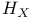 225 226 225 226
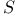 matrix 471 matrix 471
 -space (gas-space) definition 80 81 -space (gas-space) definition 80 81
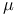 -space (molecule-space) definition 80 -space (molecule-space) definition 80
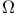 -integrals 393 422 -integrals 393 422
13 moment equations 271 273 276
Absolute temperature scale 2 3
Accommodation coefficients 278
Accommodation coefficients, describing boundary substance 242
Activity coefficient 18 64
Activity differences 161 164
Adiabatic coefficient of bulk compressibility 9
Adiabatic ellipse 570
Adiabatic Joule — Thomson coefficient 21 38 40
Adsorption 632
Adsorption spectrometer of Hagstrum 628
Air, liquified 27
Alphatron 517 538 539
Amagat units 28 29
Amagat’s law of additive volumes 58 62
Angular momentum functions, unitary transformation 473 485
Angular momentum of the molecule 469 489 491
Angular momentum of the molecule, isotropic orientation 489 491
Anisotropic Maxwellian expansion 268
Approach to equilibrium 219 225
Atomic gases, energy levels 101
Avogadro’s hypothesis 1
Back-diffusion of oil vapor 649
Backstreaming in pumps 630
Baffle 603—604
Balance, cyclic 360
Balance, detailed 360 488 490 492
Barnett effect 302 489
Barometric equation 363
Bayard and Alpert ionization gauge 612—615
Beattie — Bridgeman equation 54 55 65
Benedict — Webb — Rubin equation 54 55 56 65
Berthelot’s equation 54
Binary collisions 233 237
Binary mixtures 60
Binary mixtures, critical region 65—70
Binary mixtures, diffusion and heat conduction 412 415 423
Binary mixtures, viscosity 47 451
Binary thermodiffusion factor 412 425
Bogolubov’s derivation of the Boltzmann equation 231
Boltzmann equation, approximate 385 402 403
Boltzmann equation, as integral equation 244 245
Boltzmann equation, asymptotic solutions 253 262—266
Boltzmann equation, derivation 216 217 229—233
Boltzmann equation, for a gas of rotating molecules 481 484
Boltzmann equation, for monatomic gas mixtures 348
Boltzmann equation, for monatomic gases 347
Boltzmann equation, linear approximation 364
Boltzmann equation, linearized 244 281
Boltzmann equation, linearized, eigentensor expansion 378 381
Boltzmann equation, modifications 284—286
Boltzmann equation, normal solutions 252 265
Boltzmann equation, relativistic 345
Boltzmann equation, solubility conditions 386 404 405
Boltzmann equation, solutions in the large 249 251
Boltzmann equation, space dependent solutions 247 249
Boltzmann equation, time-independent solutions 246
Boltzmann equation, validity 205 214
Boltzmann gas 213 214 216
Boltzmann H-function see H-function
Boltzmann limit 79 85
Boltzmann statistics 454 457
Booster 515 547 559 560
Born and Green’s derivation of the Boltzmann equation 229
Bose — Einstein condensation 178 179 201
Bose — Einstein perfect gases 174seq.
Bose — Einstein statistics 81 454 458
Boundary conditions, 13 moment equations 276
Boundary conditions, asymptotic 265
Boundary conditions, diffuse 242
Boundary conditions, Euler equations 258
Boundary conditions, for a Knudsen gas 241 245
Boundary conditions, Navier — Stokes equations 277
Boundary conditions, normal solutions 253 258 264
Boundary conditions, polynomial expansions 277
Boundary conditions, specular 242 278
Boundary conditions, stochastic 242
Boundary layer 253 263 265 288
Boyle points 4 20 22 23
Boyle temperature 22
Boyle — Charles law 1
Bridge method 313
Bubble point 66 67
Buckingham — Corner potential 151
Burnett’s method for compressibility measurements 33 36 371 422
Cailletet — Mathias law 43 44
Calibration curves for double quartz-thread pendulums 529
Calibration curves for radiometer-vacuummeters 532
Calibration curves for the alphatron 539
Calibration of an ionization gauge 536 602 619—620
canonical ensemble 74 79
Centrifugal compressor 559 563
Chapman — Enskog , compatibility condition 260
Chapman — Enskog expansion 253 259 262 384
Chapman’s 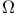 -integrals 393 422 -integrals 393 422
Characteristic line diagram after Prandtl and Busemann 569—571
Chemical bond 338
Chemical equilibrium in real gases 64 65
chemical potential 73
Chemical potential of a gas mixture 61
Clapeyron’s equation 37
Claude cycle 26 27
Clausius virial 1 0
Cluster functions 125
Cluster functions for two molecules 154
Cluster integrals 126 148
Clusters of molecules 125 131—134
Coarse graining see Smoothing
Collision cross section, differential 336 338
Collision integrals 270 271 275
Collision invariant 351 354 370
Collision operator, eigenfunctions 367 69
Collision operator, linearized 366
Collision parameter, classical mechanics 342
Collision, inverse 346
Compatibility condition, Chapman — Enskog expansion 260
Compatibility condition, Hilbert expansion 255
Compressibility factor 4 5 50 51
Compressibility of gases, measurements at high pressures 30 34
Compressibility of gases, measurements at low pressures 34 37
Compression gauge 517 518
Compression gauge after McLeod 520bis. 522
Compression gauge, turnable 522 523
Condensation 37 42 126 133 141 146
Condensation, retrograde 66 67 68
Conservation equations (mass, momentum, energy) 238 239
Conservation laws for gas mixtures 353
Conservation laws for pure gases 350
Contact gettering 637
Continuity equation 298 351 354
Cooling methods 319
Copper foil trap 656
Corresponding states for the transport coefficients 397
Corresponding states, cylindrical 292
Corresponding states, plane 290
Corresponding states, principle 48 51 62 98
Couette flow 313
| Crack products in diffusion pumps 601bis. 604
Critical constants, measurement 42 45
Critical density 42 43 141—146
Critical point 37 42 141—146
Critical ratio (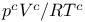 ) for several substances 49 50 ) for several substances 49 50
Critical region for binary mixtures 65 70
Critical state in a valve 567 568
Critical temperature 42 141 146
Critical volume 44
Cyclic balance 360
Dalton’s law of partial pressures 58 62
Damped oscillation, method for viscosity measurements 314
Dead space 547 550
Dead-weight gauge see Piston gauge
Degrees of freedom, separation 85 91
Density fluctuations near the critical point 46 48
Density matrix 476
Density matrix for perfect gases 192 196
Density matrix with a potential 196 201
Density matrix with a potential, symmetrization 201 204
Density matrix, general formulation 182 192
Density, critical 42 43 141—146
Density, orthobaric 43 45 53
Desorption at very low pressure 638 642
Detailed balance 360 488 490 492
Detection of leaks 541 542 661
Dew point 66 67
Diatomic molecules, rotation and vibration levels 101 108
Dieterici’s equation 54
Differential collision cross section 336 338
Diffusion coefficient of the Lorentz gas 417
Diffusion coefficient, dependence on mixing ratio 321
Diffusion coefficient, dependence on the molar fraction 431
Diffusion coefficient, determination from the formation period of thermodiffusion 325
Diffusion coefficient, first approximation 422
Diffusion coefficient, measurements 320 326
Diffusion coefficient, second approximation 432
Diffusion equation 303 408
Diffusion in a binary mixture 412 415 423
Diffusion in a ternary mixture 423
Diffusion of helium through glass 634
Diffusion pump 515 547 559
Diffusion pump, theory 576 590
Diffusion rate 357 403 407 411
Diffusion, quasi-stationary 320 321
Diffusion, representation of the cross section by scattering phases 458 461
Diffusion, stationary 322
Diffusion, thermal effect 304 323 325 330 334
Dipole gases 500
Dipole-dipole interaction, induced 339
Discharge gettering 638—640
Distribution function 152 161 345
Distribution function 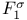 209 211 209 211
Distribution function 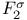 209 209
Distribution function 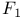 208 212 208 212
Distribution function 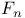 207 212 207 212
Distribution function 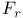 208 210 208 210
Distribution function, eigentensor expansion 390
Distribution function, locally Maxwellian 240 241
Distribution function, number density 219
Distribution function, probability density 218
Distribution function, reduced 208
Distribution function, series expansion 419
Distribution function, truncated 208
Distribution functionof the Maxwellian gas 391
Distribution matrix 476 486
Double molecule formation 311
Double quartz-thread pendulum after Coolidge and Br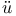 che 528 che 528
Ehrenfest — Oppenheimer law 458
Eigentensor expansion 378 381 390
Eigentensors of the Maxwellian gas 390
Eigentensors of the Maxwellian gas mixture 419
Elastic collision, equations 334 336
Electronic excitation of monatomic gases 101
Empirical equation of state 48 57
Empirical equations of state of mixtures 65
energy equation 351 355
Enskog’s approximation 384
Enthalpy 300
entropy 359
Entropy density 487
Entropy production 496
Entropy production, local 414
Entropy temperature diagram 26
Equation of continuity 298 351 354
Equation of motion 298 343 351 355
Equation of state 48 57 65
Equilibrium constant 64
Equilibrium, chemical, in real gases 64 65
Eucken relation 499—501
Euler equations 252 216
Evapor-ion pump 646
Evaporation rate 541
Evaporation, retrograde 66 67
Excess (non ideal) free-energy of mixing 61
Exchange reactions in the thermodiffusion column 329
Exhausting power 545
Exhaustion rate of diffusion pumps, von 584 590
Exhaustion rate, definition 545
Exhaustion rate, measurement 546
Expansion coefficients of a gas 5 8 9
Expansion rate of a Laval valve 568
Exponential potential model 341 397
Fermi — Dirac perfect gases 174seq. 179 182
Fermi — Dirac statistics 81 454 458
Field emission microscope 610
Flash-filament method 627
Flow birefringeance 302 489
Flow type pumps 559—605
Fluctuations in density near the critical point 46 48
Fluid blast pump 559 563 565
Fluid gauge 517 519—520
Fokker Planck equation 233
Free path 295 307 395 417
Friction coefficient, 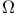 -integral representation 392 394 -integral representation 392 394
Friction gauge 517 527—530
Fugacity 17 18 21 64
Gaede’s mol-vacuummeter 533
Gaede’s vacuscope 523
Gas analysis 319
Gas constant 26 363
Gas crystal transitions 170 172
Gas degeneration 345
Gas evolution in high vacuum 631 636
Gas mixtures 57seq.
Gas mixtures, critical region 65—70
Gas mixtures, empirical equations of states 65
Gas mixtures, Perfect 57 59
Gas mixtures, real 59 64
Gas-gas immiscibility 69 70
Gases of excitable molecules 490 497
Gases of rough spheres 497—499
Gettering 636 640
Gibbs free energy 73
Grad’s method of moments 266 268 276
grand canonical ensemble 74
Haber Kerschbaum equation 528
Hagen — Poiseuille law 606
Hagstrum adsorption spectrometer 628
Half-range expansion of the distribution function 267 284
Heat capacity at constant pressure 7 28
Heat capacity at constant volume 7 28 47
Heat conduction at different gas pressures in vacuo 523 525
Heat conduction by convection 317
Heat conduction for a gas of rough spheres 498
Heat conduction in a binary mixture 412 415 451
Heat conductivity at low temperatures 467
Heat conductivity coefficient for polyatomic gases 495
Heat conductivity coefficient of the Lorentz gas mixture 418
Heat conductivity coefficient of the Maxwellian gas 383
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



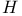 -function
-function 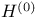
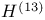
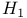
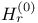
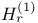
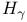
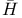
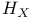
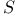 matrix
matrix  -space (gas-space) definition
-space (gas-space) definition 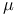 -space (molecule-space) definition
-space (molecule-space) definition 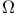 -integrals
-integrals 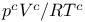 ) for several substances
) for several substances 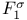
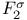
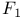
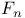
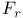
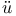 che
che