Авторизация
Поиск по указателям
Winterbone D.E. — Advanced thermodynamics for engineers
Обсудите книгу на научном форуме Нашли опечатку?
Название: Advanced thermodynamics for engineersАвтор: Winterbone D.E. Аннотация: Although the basic theories of thermodynamics are adequately covered by a number of existing texts, there is little literature that addresses more advanced topics. In this comprehensive work the author redresses this balance, drawing on his twenty-five years of experience of teaching thermodynamics at undergraduate and postgraduate level, to produce a definitive text to cover thoroughly, advanced syllabuses.
Язык: Рубрика: Физика /Термодинамика, статистическая физика /Статус предметного указателя: Готов указатель с номерами страниц ed2k: ed2k stats Год издания: 1997Количество страниц: 378Добавлена в каталог: 09.10.2005Операции: Положить на полку |
Скопировать ссылку для форума | Скопировать ID
Предметный указатель
8 101 105 acetylene 185 217 300 Acid rain 286 Activation energy 279—285 311 Activity coefficient 353 Adiabatic combustion 187—205 Adiabatic compressibility 115 Adsorption, gas molecules 294 Air 152 159 Air standard cycle 71 189 208 Air, composition 159 Air-fuel ratio 30—31 186—188 225 265—272 292 Air-fuel ratio, lean(weak) 186 223 245 265—272 301 Air-fuel ratio, rich 186 222 224 245 252 265—272 301 Air-fuel ratio, stoichiometric 186 216—217 233 267 269 299 Alcohols 185 213 217 Alkanes 138 185 217 Alkenes 185 217 Amagat's Law 121—122 Ammonia 296 Amount of substance 7 100 158—159 172—177 189 196—205 219—259 268 275 319 348 Annular combustion chamber 313 Aqueous solution 347—353 Argon 159 Aromatics 217 Arrhenius equation 279 294 299 311 Atmospheric nitrogen 159 Atomic nitrogen 242—245 273 282 Atomic oxygen 164 242—245 273 282—284 Atomic weight 158 Atomisation 211 Atomisation energy 208—215 Availability 13—36 64 359 Availability balance 27—36 Availability transfer 29 Availability, change of 18 22—29 Availability, closed system 18 27—30 Availability, effect of change of entropy 32 Availability, effect of change of volume 30 Availability, effect of combustion 30—34 Availability, flow 21 35 Availability, loss of 13—36 Availability, molar 15 Availability, non-flow 15 18 30 Availability, open system 34—36 Availability, rate of change 29 35 Availability, reaction 31 40—41 Availability, specific 15 19 27—29 Availability, steady flow 21 35 Availability, work 29 Available energy 21—26 36 67 Avogadro's hypothesis 159 Avogadro's number 340 347 Barrel swirl (tumble) 307 Baruah 268 Beattie — Bridgeman equation 128 Bejan 85 Benson 164 benzene 202 209—210 217 225 Benzoic acid 215 Bertholet equation 129 Binary cascade cycle 137 Boiler 276 291 Boiling point 137 Boltzmann constant 340 Bond energy 40 182 187 208—217 291 348 Bond energy, atomisation energy 209—217 Bond energy, dissociation energy 209—217 Bond energy, minimum potential energy 208—211 Bosch Smoke Number 287 Boyle's law 121—122 Bradley 303 Brake thermal efficiency 304 Branching reactions 295—296 Bronchial irritants 286 Bunsen burner 297 305 Butane 135—136 185 192 217 296 Calorific value 64 Calorific value, higher 188—205 Calorific value, lower 188—205 Carbon dioxide 9 40 135 139—140 159 164 168 182—205 209 211 218—275 292 345 Carbon monoxide 1 9 40 159 164 168 184—192 218—275 285 288 296 300 Carbon particulates 285 287 312 Carbon-12 158—159 192 Carbon/hydrogen ratio 185 224 270 272 Carnot cycle 21 65 68 85 89 Carnot efficiency 65—82 85—96 345 Cascade 137 Cascade, binary cycle 137 Cascade, ternary cycle 137 Catalytic converters 288 Cetane 217 Cetane number 311 Chain reaction 295—296 Charles' law 121—122 Chemical bonds 187 208—217 354 Chemical composition 186—188 196—206 229—238 245—257 265—285 Chemical equilibrium 218—257 Chemical kinetics 8 194 245 265 275 276—289 295 299 306 chemical potential 7 8 100 165 220—231 319 332 334—337 349—353 Chemical potential, standard 227—230 Chemical reaction 185—285 Chemical structure of fuels 208—217 Chemistry, combustion 184—187 208—217 Chlorine 351 Clapeyron equation 117 Clausius equation of state 123 Clausius inequality 14 Clausius — Clapeyron equation 115 117 Closed system 5—7 13—21 27—34 218 Coal 185 Coefficient of expansion 114 123—125 144 Coefficient of performance 137—138 Cold utility 47—60 Combined cycle gas turbine (CCGT) 47 92 287 Combined cycle heat engine 92—96 137 Combustion 182—313 Combustion chamber 307—313 Combustion systems, diesel 309—312 Combustion systems, gas turbine 312—313 Combustion systems, spark ignition 307—309 Combustion with heat transfer 194 199 Combustion with work transfer 194 Combustion zones, gas turbine 313 Combustion, adiabatic 187—274 Combustion, chemistry 184—187 208—217 Combustion, constant pressure 193 202 312 Combustion, constant volume 193 196 199—202 233—238 245—257 267—275 Combustion, diffusion 291 Combustion, energy equation 188—205 245—257 Combustion, exergy change 40 Combustion, heat release 208 312 Combustion, heterogeneous 183 292 305—306 Combustion, homogeneous 183 292 296—305 Combustion, incomplete 195 204 233—238 245—257 Combustion, initiation 304—305 310 Combustion, laminar 183 296—302 Combustion, multi-phase 183 Combustion, non-premixed 183 305—306 309—313 Combustion, premixed 292—305 307—309 Combustion, rich mixture 186 195 202—205 224 233—238 252—254 267—274 Combustion, single-phase 183 Combustion, speed of 297—304 Combustion, three dimensional 183 Combustion, turbulent 183 302—303 Combustion, two-dimensional 183 Combustion, weak mixture 186 200 223 245—252 267—274 Combustion, zero-dimensional 183 Composite diagram 55 60 Composite temperature - heat load diagram 51 57 Compressibility, adiabatic 115 Compressibility, factor 129 Compressibility, isentropic 115 Compressibility, isothermal 114—115 125 Compression coefficient 129 Compression, isentropic 15—18 24—25 65—82 Compressor 24—25 76—82 Concentration component 319 Concentration gradient 316 332 335 360 Concentration, molar 277—284 Condenser 69—76 Conductance, heat transfer 92—95 Conduction of electricity 318 322—332 Conduction of heat 317 319—332 Conjugate fluxes 319—342 Conjugate forces 319—342 copper 347—351 Copper sulfate 347—351 Corresponding states, law of 125—129 Coulombic forces 348 Counterflow heat exchanger 37—40 coupled equations 316—342 Coupled processes 319—342 Coupling matrix 319—342 Critical isotherm 125—127 Critical point 124—131 135 Critical point, water 126 135 Critical pressure 124 135 140 Critical temperature 124 135 146 Critical volume 124 Cross-coupling 316—342 Crude oil 184 Current, electric 316 322—332 347—361 Current, electric, density 321—332 Cycle calculations 30—34 48 69—82 Cycle efficiency 69—82 Cycles, air standard 71 189 208 Cycles, bottoming 34 Cycles, Carnot 21 65 68 85 89 Cycles, Diesel 68 Cycles, endo-reversible 65—82 85—96 Cycles, gas turbine, see Joule Cycles, internally reversible 65—82 85—96 Cycles, irreversible 65—82 Cycles, Joule 68 76—82 Cycles, Otto 30—34 68 267 Cycles, Rankine 69—76 79 Cycles, reversible 65 Cycles, Stirling 85 Cyclone separator 288 Dalton Principle 172 Damkohler 302 Daniell cell 345—351 Datum pressure, also standard pressure 164 228—229 de Groot 333 dead state 14—43 65—82 Dead state, pressure 14—43 Dead state, temperature 14—43 65—82 Debye theory 162 Deflagration 183 296—305 Delocalised electrons 210 Desorption, gas molecules 294 Destruction of availability 24—29 39—41 Detonation 183 296 309 Diatomic 184 Diesel cycle 68 Diesel engine 309—312 Diesel oil 217 Dieterici equation of state 129 Diffuser 313 Diffusion 175 294 319 332—339 359 Diffusion, burning 287 305—306 311 Diffusion, flames 291 297 305—306 Diffusivity mass 298 332—342 Diffusivity, thermal 298 303 332—342 Dilution zone 313 Direct injection engines 309—312 Dissociation 7 182 194—195 265—275 Dissociation energy 208—210 214 Dissociation, degree of 218—259 Dissociation, effect of pressure 239—240 Dissociation, effect of temperature 239—241 Dry ice (carbon dioxide) 139 Dryness fraction (quality) 137 150 Dufour effect 332—338 Dynamic equilibrium 218 222 349 Eddy diffusivity 302 Efficiency at maximum power 85—96 Efficiency, cycle 64—82 Efficiency, fuel cell 357—359 Efficiency, isentropic 22—25 30—34 69—82 Efficiency, overall 64 Efficiency, rational 65—82 154 359 Efficiency, thermal 17 64—82 85—96 292 355 Electrical cells 346—351 Electrical conduction 316 323—332 345—361 Electrical flow 323—332 Electrochemical potential 349—361 Electrode 346—351 Electrostatic precipitator 288 Elementary reaction 278 End gas 309 Endoreversible heat engine 85—96 Endothermic reaction 184 256 273—275 295 energy equation 5—6 14—15 27 34 58 86 143 189—205 233—257 347—349 Energy equation, steady flow 58 89 143 189—205 Energy equation, unsteady flow 34 Energy, available 13—36 Energy, Gibbs see Gibbs energy Energy, Helmholtz see Helmholtz energy Energy, internal 5—9 13—34 37—40 100—118 160—178 187—205 245—257 Energy, potential 1—4 Energy, unavailable 6 21—22 26 37 67 80 Engine, combustion systems 307—313 Engine, diesel 276 286—287 291 305 309—312 Engine, irreversible 65 Engine, petrol see spark-ignition Engine, reversible 5 17 21 26 65 85—96 Engine, spark-ignition 208 276 291 304 307—309 Engine, stratified charge 305 Enthalpy 6—9 22—25 34—36 40—43 100—118 141—154 160—175 187—205 245—257 Enthalpy coefficients of gases 167—170 Enthalpy of formation 65 187—205 209—217 241—242 Enthalpy of products 188—205 245—257 Enthalpy of reactants 188—205 245—257 Enthalpy of reaction 40—41 64 187—217 355—361 Enthalpy, molar 160 165 174 Enthalpy, specific 160 337 Enthalpy-temperature diagram 188—205 entropy 3—7 19—43 64—82 91 100—118 162—178 218—221 316—342 Entropy change 3 19 23 28 37 39 117 348 Entropy change due to mixing 175—178 229—231 Entropy creation 3—4 317—322 333 336 Entropy flow 5 317—322 Entropy flux 321—336 Entropy generation see creation Entropy mixtures 175—178 229—231 Entropy of transport 324—332 Entropy production see creation Entropy, ideal gas 23—25 77—82 162 165—166 Entropy, maximum 4 319 Environment 23 see Equation of constraint 227 Equations of state 121—131 158—159 Equations of state, Beattie — Bridgeman 128 Equations of state, Bertholet 129 Equations of state, Dieterici 129 Equations of state, ideal gas 105—107 110 121—123 144 159—178 195—205 233—238 Equations of state, perfect gas see also ideal gas 15—19 30—34 76—82 Equations of state, van der Waals' gas 123—128
Реклама
 |
|
О проекте
|
|
О проекте



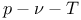 diagram
diagram