|
|
 |
| Авторизация |
|
|
 |
| Поиск по указателям |
|
 |
|
 |
|
|
 |
 |
|
 |
|
| Weissbluth M. — Atoms and Molecules |
|
|
 |
| Предметный указатель |
Magnetic field interaction, with spin-orbit coupling 350—354
Magnetic field interaction, Zeeman levels 349—350
Magnetic hyperfine interaction 358—374
Magnetic hyperfine interaction, and magnetic field 371—374
Magnetic hyperfine interaction, coupling tensor 367
Magnetic hyperfine interaction, Fermi contact term 367
Magnetic hyperfine interaction, Hamiltonian 358—367
Magnetic hyperfine interaction, one-electron system 367—374
Magnetic moment, gyromagnetic ratio 367
Magnetic moment, operators 347—349
magnetization 277—278
Matrix element, reduced 159 162—163
Matrix element, theorem 88—90 103—104
Molecular orbitals see also Molecules
Molecular orbitals, angular momentum properties 577—578
Molecular orbitals, antibonding 600
Molecular orbitals, bond order matrix 599
Molecular orbitals, bonding 600
Molecular orbitals, CNDO (complete neglect of differential overlap) 563
Molecular orbitals, computational methods 561—565
Molecular orbitals, EHM (extended Hvickel method) 564
Molecular orbitals, H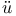 ckel method 597—603 ckel method 597—603
Molecular orbitals, Hartree — Fock 556—560
Molecular orbitals, inversion symmetry 578
Molecular orbitals, L wdin orbitals 564—565 wdin orbitals 564—565
Molecular orbitals, LCAO (linear combination of atomic orbitals) 556
Molecular orbitals, NDO (neglect of differential overlap) 562
Molecular orbitals, nonbonding 594 600
Molecular orbitals, pi ( )-electron approximation 596—603 )-electron approximation 596—603
Molecular orbitals, PPP (Pariser — Parr — Pope) method 604—605
Molecular orbitals, Roothaan method 556—560
Molecular orbitals, sigma ( ) and pi ( ) and pi ( ) bonds 574—576 ) bonds 574—576
Molecular spectra 606—649
Molecular spectra, diatomic molecules 615—631
Molecules 551—649 (see also Bom — Oppenheimer approximation; Diatomic molecules; Hydrogen molecule; Hydrogen molecule ion; Molecular orbitals; Molecular spectra)
Molecules, benzene 600—603
Molecules, bonds 575—576 587—589 594 600
Molecules, butadiene 597—600
Molecules, carbon dioxide 590—591
Molecules, electronic states 566—605
Molecules, ethylene 604—605
Molecules, formaldehyde 595—596
Molecules, hybrid orbitals 591—596
Molecules, oxygen 585—587
Molecules, polyatomic 631—641
Molecules, polyenes 602
Molecules, population analysis 588—589
Molecules, vibrations 606—609 611 617 635—641
Molecules, water 594—595
Morse potential 608—609 (see also Diatomic molecules)
Multielectron atom 385—481 (see also Hartree — Fock formulation; Multiplet)
Multielectron atom, 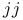 coupling 441—442 coupling 441—442
Multielectron atom, closed shell 392
Multielectron atom, configuration 392
Multielectron atom, electric field interaction 479—480
Multielectron atom, equivalent electrons 392
Multielectron atom, Hamiltonian 385—389
Multielectron atom, levels 393
Multielectron atom, LS (Russell — Saunders) coupling 388
Multielectron atom, magnetic field interaction 478—479
Multielectron atom, parity 391
Multielectron atom, terms 393
Multiplet 413—442 (see also Multielectron atom)
Multiplet Fluorescence 530 647—648
Multiplet, coefficients of fractional parentage (cfp) 430—435
Multiplet, equivalent electrons 421 425
Multiplet, group properties 435—441
Multiplet, Hund’s rules 426 586
Multiplet, inverted 478
Multiplet, Land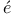 interval rule 467 472 interval rule 467 472
Multiplet, multiplicity 415
Multiplet, normal 467
Multiplet, parity 425
Multiplet, regular 477
Multiplet, seniority number 426
Multiplet, terms from equivalent electrons 418—420
Multiplet, two-electron 413—420
Multiplet, wave functions 427—435
Normal coordinates 633
Number operator 260
Occupation number space see Fock space
One-electron, matrix elements 454—457
One-electron, spin orbital 335
One-electron, wave functions 334—337
Optical susceptibility 547—550
Optical susceptibility, nonlinear effects 548—550
Orbital angular momentum 1—3 8-9
Orbital angular momentum, commutation rules 2
Orbital angular momentum, eigenvalues and eigenfunctions 8—9
Oscillator strengths 512—515
Oscillator strengths, sum rules 513—514
Pauli principle 257—258 288
Permutation groups 143—150 435—441
Permutation groups, basis functions 148—150
Permutation groups, Cayley’s theorem 145
Permutation groups, irreducible representations 147
Permutation groups, Young diagrams 145—147
Permutation groups, Young operator 148—150
Perturbations, Brillouin — Wigner expansion 296
Perturbations, Fermi’s golden rule 299—301
Perturbations, harmonic potential 298—299
Perturbations, random 301—305
Perturbations, Rayleigh — Schrodinger expansion 296
Perturbations, time-dependent 297—299 (see also Evolution operator)
Perturbations, time-independent 294—297
Phosphorescence 648
photoelectric effect 521—525
Planck’s radiation law 511—512
Plane wave expansion 193—196
Plane wave expansion, multipoles 199—202
Point groups, character tables 115—120
Point groups, classification 114
Point groups, coupling coefficients 132—142
Point groups, double (extented) groups 121—127 140
Point groups, nomenclature 112—113 124 127 142
Point groups, symmetry operations 111—112 124 127 142
Polyatomic molecules, electronic transitions 641
Polyatomic molecules, Raman scattering 644—646
Polyatomic molecules, relaxation 646—649
Polyatomic molecules, vibrational transitions 642—643
Polyatomic molecules, vibronic interaction 643 644
Quadrupole interaction see Electric quadrupole interaction
Quantum mechanics and group theory 203—207
Quenching see Ligand fields
Racah parameters 249
Racah W coefficients 39 (see also Six 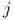 (6 (6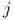 ) symbols) ) symbols)
Radiation and atoms see also Absorption and emission; Electromagnetic interactions
| Radiation and atoms, angular distribution 503—504
Radiation and atoms, electric dipole 494 497—500
Radiation and atoms, electric quadrupole 496—497 500
Radiation and atoms, Hamiltonian 492—493
Radiation and atoms, magnetic dipole 495—496 500
Radiation and atoms, matrix elements 493—494
Radiation and atoms, nonlinear effects 309—310 547—550
Radiation and atoms, selection rules 500—502
Radiation field see also Radiation and atoms
Radiation field, box normalization 483—484
Radiation field, creation and annihilation operators 488—490
Radiation field, Hamiltonian 483—488
Radiation field, photon description 489—490
Radiation field, polarization 484—485
Radiation field, quantization 488—492
Radiation field, rotational properties 491
Radiation field, transversality 484
Radiation field, zero point energy 490
Raman scattering 644—646 (see also Polyatomic molecules)
Rayleigh scattering 544
Rayleigh — Schr dinger expansion 296 dinger expansion 296
Reaction matrix 232
Reduced matrix element see Wigner — Eckart theorem
Relaxation, molecular 646—649
Response function 305—307 309—312
Roothaan method 556—560 (see also Molecular orbitals)
Rotation groups 91—110
Rotation groups, relation between 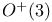 and SU(2) 98—100 and SU(2) 98—100
Rotation groups, three-dimensional 93 95 100—103
Rotation groups, two-dimensional 91—93
Rotational transitions, diatomic 618—620
Rotations see also Rotational transitions; Rotation groups
Rotations, and angular momenta 51 53
Rotations, diatomic molecules 609—615
Rotations, Euler angles 54—55
Rotations, Hund’s cases 614—615
Rotations, transformation of angular momentum eigenfunctions 55—59
Scaling 572 (see also Virial theorem)
Scattering, Kramers — Heisenberg formula 536—542
Scattering, matrix elements 230—232
Scattering, operator 228
Scattering, Raman 542—544 644—646
Scattering, Rayleigh 542—544
Scattering, Thomson 542
Schr dinger representation 216—220 dinger representation 216—220
Selection rules see also Wigner — Eckart theorem
Selection rules, absorption and emission 509
Selection rules, by group theory 89—90 103—104
Selection rules, diatomic molecules 627—631
Selection rules, Raman 645—646
Selection rules, rotational 617—619
Selection rules, three-dimensional rotation group 103 104
Selection rules, vibrational 617
Seniority number 426
Six 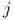 (6 (6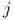 ) symbols 38—44 ) symbols 38—44
Six 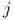 (6 (6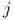 ) symbols, numerical values 43—44 ) symbols, numerical values 43—44
Six 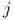 (6 (6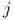 ) symbols, special formulas 40—42 ) symbols, special formulas 40—42
Slater determinant 215 240—253 391 395
Slater determinant, closed shell 251—253
Slater determinant, matrix elements 240—243
Slater orbitals 409
Slater — Condon parameters 247—249 (see also Coulomb interaction)
Spectra see Atomic spectra; Molecular spectra
Spherical harmonics 3—12
Spherical harmonics, Condon — Shortley convention 5
Spherical harmonics, Gaunt formula 12
Spherical harmonics, real combinations 6
Spin, density 271—273 287
Spin, eigenvalues and eigenfunctions 20—22 46
Spin, Hamiltonian 675—677 (see also Ligand fields)
Spin, matrix elements 20—22 45
Spin, orbital 335
Spin, Pauli matrices 22
Spin, under rotation 107
Spin, undertime reversal 208—210
Spin-orbit interaction 337—341 465—476
Spin-orbit interaction, and magnetic fields 350—354
Stark effect 479—480
susceptibility 306—312
Susceptibility, energy dissipation 306—307
Susceptibility, Kramers — Kronig relations 307—309
Susceptibility, nonlinear 309—310
Susceptibility, quantum mechanical 311—312
Symmetry see also Groups
Symmetry, diatomic molecules 622—631
Symmetry, hydrogen molecule ion 573—579
Symmetry, of the Hamiltonian 203—215
Symmetry, reduction due to a perturbation 204—207
Tensors see also Irreducible tensor operators
Tensors, basis for permutation groups 435—441
Tensors, cartesian 167—175
Tensors, product 153 155—159
Terms see also Diatomic molecules; Multiplet
Terms, equivalent electrons 418—420
Terms, seniority number 426
Three 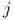 (3 (3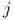 ) symbols 12 31—35 ) symbols 12 31—35
Three 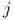 (3 (3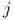 ) symbols, numerical tables 34—35 ) symbols, numerical tables 34—35
Three 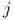 (3 (3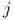 ) symbols, special formulas 33 ) symbols, special formulas 33
Three 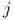 (3 (3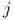 ) symbols, triangle condition 12 ) symbols, triangle condition 12
Time reversal 207—213 (see also Kramers’ theorem)
transitions see Atomic spectra; Molecular spectra; Polyatomic molecules
Unit tensor operator 456 (see also Irreducible tensor operators)
Unit vectors 153—155 186 189 686—687
United atom treatment 579—580 (see also Hydrogen molecule ion)
Vacuum state 254
Valence bond method 583 (see also Hydrogen molecule)
Variational method 290—294
Vector fields 182—202
Vector spherical harmonics 187—193
Vibrations, diatomic molecules 606—609 611 617
Vibrations, group theory 635—641
Vibrations, Morse potential 608—609
Vibrations, normal coordinates 633
Vibrations, polyatomic molecules 631—641
Vibrations, vibronic interactions 643 644
Virial theorem 570—572
Wigner — Eckart theorem 159—167 377—379
Wigner — Eckart theorem, Land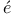 formula 348 364—367 379 formula 348 364—367 379
Wigner — Eckart theorem, reduced matrix element 159 162—163 166
Wigner — Eckart theorem, selection rules 160—161
X-rays, absorption spectra 532
X-rays, Auger effect 532—533
X-rays, fluorescence 530
X-rays, selection rules 530
X-rays, terms 530—532
Young diagrams see Permutation groups
Zeeman effect see Magnetic field interaction
|
|
 |
| Реклама |
 |
|
|
 |
|
О проекте
|
|
О проекте



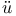 ckel method
ckel method  wdin orbitals
wdin orbitals  )-electron approximation
)-electron approximation  ) and pi (
) and pi (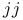 coupling
coupling 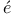 interval rule
interval rule  (6
(6 and SU(2)
and SU(2)